6.2 Properties of Transition Elements
The transition elements exhibit distinct characteristics that set them apart from other elements in the periodic table. Unlike the gradual change from metals to non-metals observed across Period 3, transition elements are all metals. They also display less significant decreases in atomic radii across a period compared to main group elements. These similarities give rise to their unique physical and chemical properties.
Interesting Information: The atomic radii of vanadium () and zinc () are observed to be similar. This is attributed to the shielding effect provided by the addition of electrons, which effectively counterbalances the increasing nuclear charge.
6.2.1 Physical Properties
Transition metals generally share the following physical properties:
- High Melting Points: Due to strong metallic bonding involving both and electrons.
- High Density: Resulting from the close packing of atoms in their metallic lattices.
- Good Electrical Conductors: They possess delocalized (free) electrons.
- Good Thermal Conductors.
- Shiny Metallic Luster.
- Malleable and Ductile: Can be hammered into sheets and drawn into wires.
- Hard and Strong: Making them suitable for construction and manufacturing.
- Magnetic Properties: They exhibit paramagnetism and ferromagnetism, properties exploited in applications like magnetic resonance imaging (MRI).
6.2.2 Physical Characteristics (Detailed)
This section delves deeper into some key physical characteristics of the first row of transition metals.
1. Density
Density is defined as mass per unit volume, typically expressed in . It reflects how closely metal atoms are packed in the metallic lattice.
- General Trend: Moving across the first period of transition metals (from Scandium to Copper), the density generally increases.
- Reasons for Trend:
- Increasing Atomic Mass: Nuclei become progressively heavier across the period.
- Decreasing Atomic Radius: Increasing nuclear charge pulls the electron cloud closer, reducing the atomic volume.
- Combined Effect: Since density is directly proportional to mass and inversely proportional to volume (), the simultaneous increase in mass and decrease in volume lead to an overall increase in density.
- Exceptions: Manganese and Copper show deviations from this general trend. Zinc, for instance, has a larger atomic radius than copper due to its full sub-shell, which leads to less tight packing of zinc atoms in its metallic lattice, thereby reducing its density.
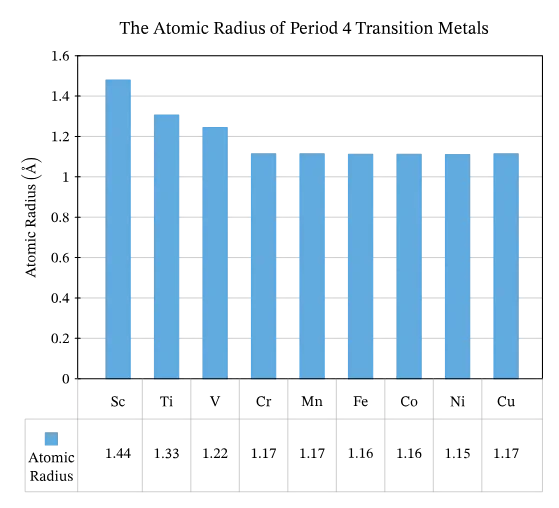
2. Atomic Radii
The atomic radii of first-row transition elements show specific trends influenced by both shielding effect and electronic configuration.
- General Trend: Atomic radii generally decrease across the period due to increasing nuclear charge pulling electrons closer.
- Less Steep Decrease: Unlike main group elements, this decrease is less pronounced because electrons added to the sub-shell do not effectively shield the outer electrons from the increasing nuclear charge.
- Early Period: Initially, atomic radii decrease because the increasing nuclear charge overcomes the shielding effect.
- Middle of the Period: The decrease in atomic radii becomes less pronounced as the increasing nuclear charge and shielding effect begin to counterbalance each other, resulting in almost constant atomic radii.
- End of the Period (Nickel to Copper and Zinc): Atomic radii increase.
- From Nickel to Copper, the sub-shell becomes full, leading to increased electron-electron repulsion and an enhanced shielding effect that pushes the sub-shell further from the nucleus.
- From Copper to Zinc, an additional electron in the sub-shell further increases the shielding effect, outweighing the effect of the higher nuclear charge, resulting in an unexpected increase in atomic radius.
Important Information: When discussing covalent and ionic radii, two factors are considered simultaneously: electron-electron repulsion and electron-nucleus attraction.
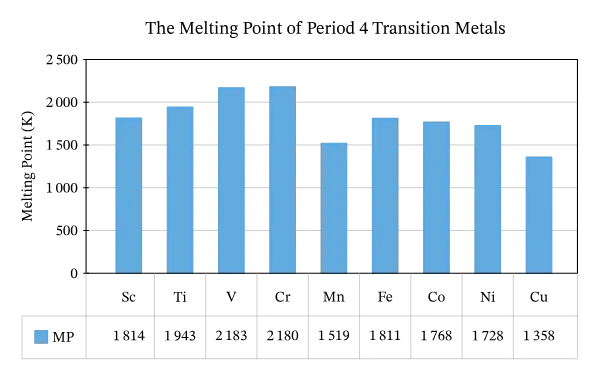
3. Melting and Boiling Points
- High Values: Transition elements generally have much higher melting and boiling points compared to s- and p-block elements.
- Reason: This is due to strong metallic bonding, which involves electrons from both the and sub-shells.
- Irregular Behavior: Plots of melting and boiling points against atomic number show irregular behavior.
- Initially, they increase with atomic number.
- They show minima at Manganese, reflecting that its half-filled sub-shell () and full sub-shell () are particularly stable and less readily available for metallic bonding.
- Zinc has the lowest melting and boiling points among the first-row transition metals because its full and sub-shells do not significantly participate in metallic bonding. This explains why mercury, belonging to the same group as zinc, is liquid at room temperature.
Key Information: Metallic bonding strength is directly related to the number of unpaired electrons in outermost shells. However, the stability of filled or half-filled sub-shells also plays a crucial role.
4. Magnetic Properties
Transition metals and their compounds exhibit diverse magnetic properties, providing insights into their chemical bonding and enabling technological applications. Electron spin generates a magnetic field, so a single unpaired electron acts like a small magnet.
- Paramagnetism:
- Description: Substances that are weakly attracted to a magnetic field.
- Cause: Presence of one or more unpaired electrons in their valence shells. These unpaired electrons on atoms or ions are randomly oriented.
- Behavior: When placed in a magnetic field, their magnetic moments align parallel to the field, causing a net weak attraction. They lose their paramagnetic properties once the external magnetic field is removed.
- Example (from text): Aluminium, Sodium (While Al and Na are not transition metals, they are often used as examples of paramagnetic substances due to unpaired electrons, though their magnetism is very weak compared to transition metals).
- Ferromagnetism:
- Description: Substances that are strongly attracted to a magnetic field (millions of times more than paramagnetic substances).
- Cause: Presence of unpaired electrons in the valence shells of their atoms or ions in the solid state. These unpaired electrons are influenced by surrounding atoms and align in the same direction, forming magnetic domains.
- Behavior: They exhibit a very strong attraction in a magnetic field and retain their magnetic moment even after the field is removed, making them permanent magnets.
- Examples: Iron (Fe), Cobalt (Co), Nickel (Ni).
- Diamagnetism:
- Description: Substances that are weakly repelled by a magnetic field.
- Cause: All electrons in their outermost shells are paired.
- Behavior: The paired electrons in an orbital have opposite spins, canceling out their individual magnetic moments, resulting in a net magnetic moment of zero.
- Example (from text): Copper (Cu), Iron (Fe - note: elemental iron is ferromagnetic, but some iron compounds can be diamagnetic).
5. Alloys
- Definition: Alloys are mixtures of metals (or metals with non-metals) that possess useful properties often superior to their constituent elements.
- Formation by Transition Metals: Transition metals can readily form alloys due to their similar atomic sizes, allowing them to substitute one another in their metallic lattices.
- Examples:
- Steel: An alloy of iron, chromium, nickel, and manganese. It exhibits enhanced properties like increased hardness, corrosion resistance, and a shiny appearance.
- Brass: An alloy of copper and zinc.
- Bronze: An alloy of copper and tin.
6.2.3 Chemical Properties
The chemical properties of first-row transition metals show less similarity compared to their physical properties.
For more on electron configurations that influence these properties, see Electron Configurations Of The D Block Elements→.
1. Variable Oxidation States
- Common Oxidation State: The most common oxidation state for the first row of -block elements is +2, which is achieved by the loss of the two electrons to form an ion.
- Maximum Oxidation State: The maximum stable oxidation state often corresponds to the total number of electrons available for bonding from both the and sub-shells. For example, manganese, with an electronic configuration of , can exhibit a maximum oxidation state of +7.
Do You Know?: Simple ions of transition metals, such as , are typically stable in low oxidation states, whereas their complex ions, like (permanganate ion), are stable in high oxidation states.
Oxidation States of First Row Transition Metals:
| Element | Common Oxidation States |
|---|---|
| Sc | +3 |
| Ti | +2, +3, +4 |
| V | +2, +3, +4, +5 |
| Cr | +2, +3, +4, +6 |
| Mn | +2, +3, +4, +6, +7 |
| Fe | +2, +3 |
| Co | +2, +3 |
| Ni | +2 |
| Cu | +2 |
| Zn | +2 |
- Stability Trends:
- The stability of the +2 oxidation state generally increases from left to right across the first row of the -block, as it becomes progressively more difficult to remove electrons.
- Conversely, the stability of the higher oxidation states generally decreases from left to right across the series.
- Half-filled Sub-shell Stability: The greater stability of the +2 oxidation state of manganese (forming with ) compared to its +3 oxidation state, and the +3 oxidation state of iron (forming with ) relative to its +2 oxidation state ( with ), can be explained by the enhanced stability of a half-filled sub-shell.
2. Catalytic Activity
Transition elements and their compounds are widely used as catalysts in various chemical reactions due to two unique properties:
-
Multiple Stable Oxidation States: They can exist in more than one stable oxidation state.
-
Vacant Orbitals: They possess vacant orbitals capable of forming temporary dative bonds with ligands.
-
Mechanism of Catalysis:
- Variable Oxidation States: First-row transition metals can readily switch between different oxidation states (e.g., and , or to ). During a reaction, they temporarily change their oxidation states by gaining or losing electrons, facilitating the conversion of reactants into products. After the reaction, they regain their original oxidation state, ready to catalyze again.
- Dative Bond Formation: The incomplete -orbitals in their valence shells allow them to accept electron pairs from ligands (molecules or ions). This forms temporary dative bonds between the transition metal and the ligands. These dative bonds are crucial for:
- Holding reactants on the surface of the transition metal, bringing them into close proximity.
- Weakening and ultimately breaking bonds within the reactant particles.
- Facilitating product formation on the metal surface.
- Once the reaction is complete, the transition metal releases the products and is regenerated, ready to bind to new reactants.
-
Examples of Catalysis:
- Haber Process: Iron (Fe) is used as a heterogeneous catalyst for the synthesis of ammonia (). It adsorbs hydrogen () and nitrogen () gases on its surface, increasing their reaction rate.
- Contact Process: Vanadium pentoxide () acts as a heterogeneous catalyst in the production of sulfuric acid. Vanadium undergoes a reversible change in its oxidation state from +5 to +4 during the reaction.
- Oxidation of Iodide Ion: The reaction between iodide ion () and peroxodisulphate (VI) ion () is typically slow due to repulsion between the negatively charged reactant ions: This reaction can be catalyzed by the addition of -block metal ions, such as , illustrating homogeneous catalysis. The mechanism involves reversible changes in the iron's oxidation state: Both steps involve reactions between oppositely charged ions, which occur much faster than the uncatalyzed reaction.
3. Ligands
A ligand is an electron-rich atom or group of atoms (molecules or ions) that can donate one or more lone pairs of electrons to a central transition metal ion, forming dative covalent bonds. Ligands are also known as Lewis bases or nucleophiles due to their electron-donating capability.
For more on how ligands affect transition metal compounds, see Formation Of Complex Compounds→ and Coloured Complexes→.
Key Information: The phenomenon of sub-shell splitting (crystal field splitting) is caused when ligands approach the central metal ion, influencing the metal's electronic and optical properties.
Ligands are classified based on the number of lone pairs they can contribute to coordinate covalent bond formation:
-
Monodentate Ligands:
- Description: Ligands that can form only one dative covalent bond with a transition metal atom/ion. The term "monodentate" means one-toothed. They may possess more than one lone pair but only donate one in bonding to a single metal center.
- Examples:
- Aqua ()
- Halo (, where X is a halogen, e.g., , )
- Hydroxo ()
- Ammine ()
- Cyano ()
-
Polydentate Ligands (Chelating Ligands):
- Description: Ligands that can donate more than one lone pair of electrons to a central metal ion, forming multiple dative covalent bonds. The term "polydentate" means many-toothed. These ligands form ring structures with the metal ion, known as chelates, which are typically very stable.
- Examples:
- Bidentate Ligands (form two coordinate covalent bonds):
- 1,2-diaminoethane (), commonly known as ethylenediamine (abbreviated as en).
- Ethanedioate (), also known as oxalato.
- Hexadentate Ligands (form six coordinate covalent bonds):
- Ethylenediaminetetraacetic acid (EDTA), or its anionic form, ethylenediaminetetraacetate. This ligand can donate six lone pairs of electrons to a central metal atom (two from nitrogen atoms and four from oxygen atoms of the carboxylate groups). Its structure is .
- Bidentate Ligands (form two coordinate covalent bonds):