6.3.3 Coloured Complexes
Transition metal complexes, both in solid and solution forms, often exhibit vibrant colours due to their unique electronic structure.
Origin of Colour
The colour observed in transition metal complexes arises from the absorption of specific wavelengths of visible light.
- Black: If all visible light is absorbed.
- Coloured: If some wavelengths are absorbed, and the remaining light is transmitted or reflected. The observed colour is the complementary colour to the absorbed light.
- White: If all visible light is reflected.
Transition metal ions are typically coloured if they possess incomplete sub-shells.
- Example: Copper(II) ions () form coloured compounds because they have an incomplete sub-shell.
- Example: Zinc(II) ions () form colourless compounds because they have a complete sub-shell.
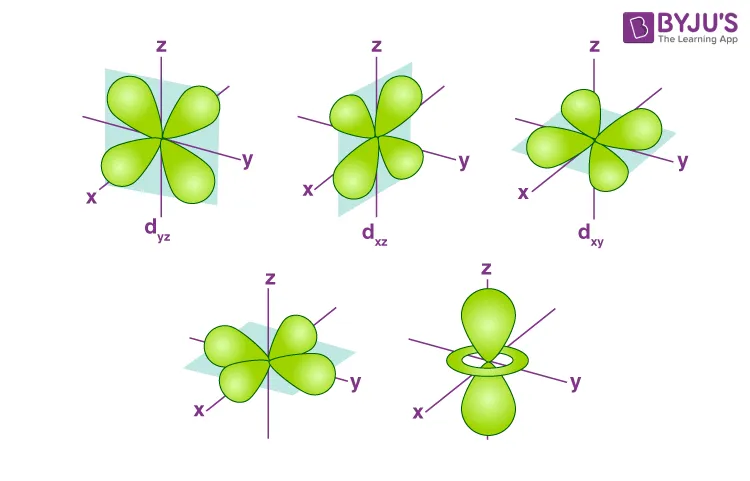
Degenerate Orbitals
All five orbitals in a free metal ion's sub-shell possess the same energy, making them degenerate orbitals. They differ in their spatial orientation along the x, y, and z axes.
Crystal Field Splitting Theory (d-d Splitting)
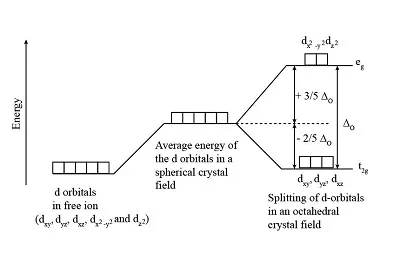
In a complex, when ligands approach the central metal ion, their electric fields interact with the metal's orbitals. This interaction lifts the degeneracy of the orbitals, causing them to split into different energy levels. This phenomenon is known as crystal field splitting.
- Octahedral Complexes: In an octahedral complex, six ligands approach the central metal ion along the x, y, and z axes.
- The and orbitals point directly along the axes towards the ligands, experiencing greater repulsion. These orbitals are raised to a higher energy level, forming the set.
- The , , and orbitals are oriented in between the axes, experiencing less repulsion from the ligands. These orbitals are lowered to a lower energy level, forming the set.
- The overall energy of the split orbitals ( and ) in the complex is higher than the degenerate orbitals of the isolated metal ion.
- Key Information:
- The terms and stand for "doubly gerade" and "triply gerade," respectively.
- The subscript "g" stands for "gerade," a German word meaning "even" (referring to the symmetry of the orbitals).
d-d Electronic Transitions and Complementary Colours
The energy difference between the and sets of orbitals is denoted as (crystal field splitting energy or ligand field splitting energy).
- When white light passes through a transition metal complex, a photon of specific energy and frequency (corresponding to ) is absorbed.
- This absorbed energy causes an electron from the lower energy orbitals to be excited to the higher energy orbitals. This process is called a electronic transition.
- The colour observed for the complex is the complementary colour to the light that was absorbed. The relationship between absorbed and observed colours is shown by the colour wheel.

- Examples:
- The nickel complex, , absorbs red light and appears green (complementary to red).
- The copper complex, absorbs orange light and appears blue (complementary to orange).
6.3.4 The d-d Splitting Pattern in Octahedral and Tetrahedral Complexes
The splitting pattern of orbitals depends on the geometry of the complex.
Octahedral Complexes
- In octahedral complexes, ligands approach along the axes.
- orbitals (, ): Experience greater repulsion from ligands because they are aligned directly towards them, leading to higher energy.
- orbitals (, , ): Lie between the ligands, experiencing less repulsion, leading to lower energy.
- Example: The aqua complex of titanium(III), . This complex absorbs yellow-green light to excite an electron from to , resulting in a red-violet colour.
Tetrahedral Complexes
- In tetrahedral complexes, ligands are positioned between the axes.
- orbitals (, , ): Point more directly towards the ligands, experiencing greater repulsion, leading to higher energy.
- orbitals (, ): Are positioned between the ligands, experiencing less repulsion, leading to lower energy.
- This is an inverted splitting pattern compared to octahedral complexes, and the splitting energy () is generally smaller than in octahedral complexes ().
- Example: Tetrachlorocuprate(II) complex, .
6.3.5 Effect of Ligands on Colour of Complexes
The nature of the ligand significantly influences the extent of -orbital splitting () and, consequently, the colour of the complex.
Ligand Field Strength
Different ligands have varying abilities to cause -orbital splitting, which is quantified by their ligand field strength.
-
Spectrochemical Series: Ligands can be arranged in a series based on their increasing ability to split orbitals:
-
Strong field ligands: Cause a larger (larger splitting), absorbing higher energy light (e.g., violet, blue) and transmitting lower energy light (e.g., yellow, orange, red).
-
Weak field ligands: Cause a smaller (smaller splitting), absorbing lower energy light (e.g., red, orange) and transmitting higher energy light (e.g., blue, green, violet).
Ligand Exchange and Colour Change
When a ligand in a complex is replaced by another ligand with a different field strength, the changes, leading to a change in the absorbed light and thus a change in the observed colour of the complex.
- Example: Addition of ammonia (a strong field ligand) to an aqueous solution of copper(II) ions (where water is a weaker field ligand). In this reaction, the water ligands are replaced by ammonia, leading to a larger splitting energy and a shift in absorption from orange (resulting in blue) to a higher energy region, making the complex appear violet-blue.
6.3.6 Geometries of Complexes
The geometry of transition metal complexes is determined by the number of ligands (coordination number) and the need to minimize repulsion between the dative bond pairs, often following VSEPR theory. However, some geometries like square planar deviate. The size of ligands can also influence the coordination number. For example, is larger than or , so fewer ions can surround a metal center.
1. Complexes with Coordination Number 6 (Octahedral)
- Shape: Octahedral.
- Ligand arrangement: Six donor atoms from ligands are positioned at the vertices of an octahedron.
- Bond angles: between adjacent ligands.
- VSEPR compliance: This arrangement maximizes distance and minimizes ligand-ligand repulsion, consistent with VSEPR theory.
- Commonality: Most common and stable complexes; almost all transition metals can form octahedral complexes.
- Ligands: Can involve both monodentate (e.g., , ) and polydentate (e.g., ethylenediamine, 'en') ligands.
- Examples:
- Hexaaquanickel(II) ion,
- Tris(1,2-diaminoethane) nickel(II) ion,
2. Complexes with Coordination Number 4
There are two main geometries for complexes with coordination number 4.
i. Tetrahedral Complexes
- Shape: Tetrahedral.
- Ligand arrangement: Four ligands positioned at the vertices of a tetrahedron.
- Bond angles: between ligands.
- VSEPR compliance: The tetrahedral arrangement maximizes the distance between ligands, consistent with VSEPR theory.
- Commonality: Less common than octahedral complexes. Often formed with chloride ligands or metal ions with electron configuration.
- Common metal ions: and .
- Examples:
- Tetrachlorocuprate(II),
ii. Square Planar Complexes
- Shape: Square planar.
- Ligand arrangement: Four ligands directed at the four corners of a square.
- Bond angles: between adjacent ligands.
- VSEPR compliance: This geometry is against simple VSEPR theory predictions (which would predict tetrahedral for 4 electron domains).
- Commonality: Less common than tetrahedral, but significant for certain metal ions.
- Common metal ions: Often formed by metal ions with electronic configuration, such as , , and .
- Examples:
- Diamminedichloroplatinum(II) (cisplatin),
3. Complexes with Coordination Number 2 (Linear)
- Shape: Linear.
- Ligand arrangement: Two monodentate ligands positioned at to each other.
- Stability: Generally less stable than other geometries.
- Examples:
- Diamminesilver(I) ion