21.2 Key Reagents and Functional Group Interconversions
This section explores essential chemical reagents used in organic synthesis, categorizing them by their primary function (oxidizing, reducing, or dehydrating agents). It also illustrates how these reagents facilitate the interconversion of various organic functional groups.
Oxidizing Agents
Oxidizing agents gain electrons and cause the oxidation of other substances. In organic chemistry, they typically increase the number of bonds to oxygen or decrease the number of bonds to hydrogen.
Acidified Potassium Dichromate () or Acidified Potassium Permanganate ()
These are strong oxidizing agents.
Primary Alcohols → Aldehydes → Carboxylic Acids:
Primary alcohols are initially oxidized to aldehydes. With excess strong oxidizing agent, oxidation proceeds further to carboxylic acids.
Example: Oxidation of ethanol:
Secondary Alcohols → Ketones:
Secondary alcohols are oxidized to ketones. Ketones resist further oxidation because the carbonyl carbon lacks a hydrogen atom.
Example:
Reducing Agents
Reducing agents lose electrons and cause the reduction of other substances. In organic chemistry, they decrease the number of bonds to oxygen or increase the number of bonds to hydrogen.
Sodium Borohydride ()
A mild, selective reducing agent. It reduces aldehydes to primary alcohols and ketones to secondary alcohols. It does not reduce carboxylic acids, esters, or double bonds.
This selectivity makes ideal when other reducible groups (e.g., alkenes) must remain intact.
Lithium Aluminum Hydride ()
A strong, versatile reducing agent. The bond is weaker and more polar than the bond in , making hydride () more available for nucleophilic attack.
Reduces: aldehydes, ketones, carboxylic acids, esters, and amides.
Hydrogen Gas with Nickel Catalyst ()
Used for catalytic hydrogenation. Reduces double bonds (alkenes → alkanes) and can also reduce alkynes.
Tin/Hydrochloric Acid ()
Used for the reduction of nitro groups to amino groups. This is the standard method for synthesizing primary aromatic amines from nitroarenes.
Example: Nitrobenzene → Aniline ()
Dehydrating Agents
Dehydrating agents remove water from a molecule. In organic reactions, they form double bonds from alcohols (elimination reactions) or facilitate condensation reactions.
Aluminum Oxide ()
Used at high temperatures for the dehydration of alcohols to alkenes.
Concentrated Sulfuric Acid ()
A strong dehydrating agent used for acid-catalyzed elimination of alcohols to alkenes and in condensation reactions.
Summary Table
| Reagent | Type | Functional Group Converted | Product |
|---|---|---|---|
| or | Oxidizing | Primary alcohol | Aldehyde → Carboxylic acid |
| or | Oxidizing | Secondary alcohol | Ketone |
| Mild reducing | Aldehyde / Ketone | Primary / Secondary alcohol | |
| Strong reducing | Carboxylic acid / Ester / Amide | Primary alcohol / Amine | |
| Reducing | Alkene () | Alkane | |
| Reducing | Nitro group () | Amino group () | |
| Conc. / | Dehydrating | Alcohol | Alkene |
Functional Group Interconversion Flowchart
The following diagram provides a visual representation of how different functional groups can be interconverted using various reagents and reaction types.
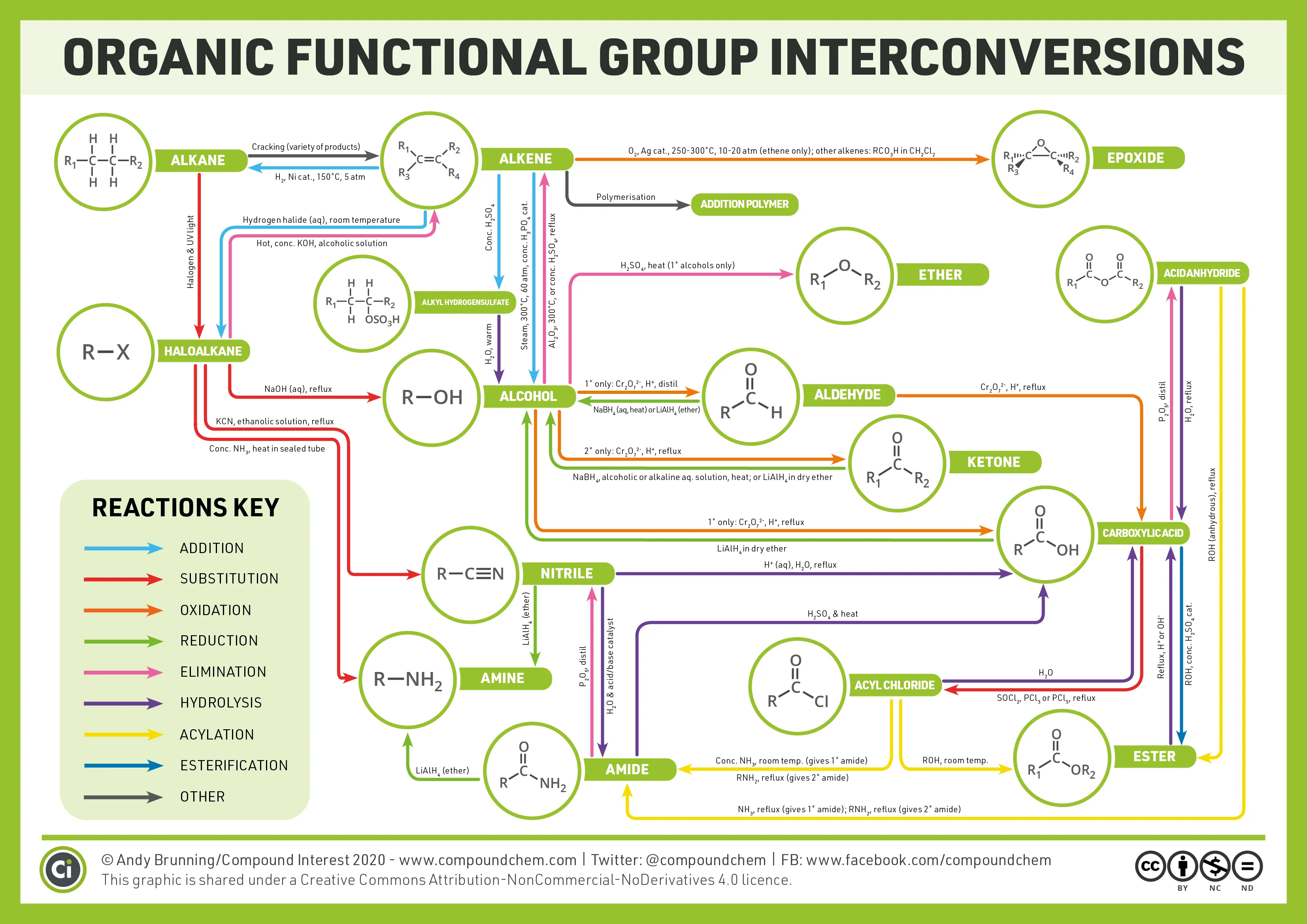
<PracticeQuestions questionsString="[{"question":"Which reagent is specifically used for the selective reduction of carbonyl groups in the presence of carbon-carbon