21.1 Important Organic Reactions
This document provides a structured overview of important organic reactions, focusing on synthesis strategies, functional group identification tests, and characteristic reactions of various homologous series. Familiarity with these reactions and their conditions is crucial for understanding organic chemistry.
Introduction to Organic Reactions
Organic chemistry involves a vast array of reactions that allow the transformation of one organic compound into another. Understanding these reactions, their mechanisms, and the specific conditions required is fundamental for successful synthesis and analysis. The flowchart below summarizes some key interconversions.
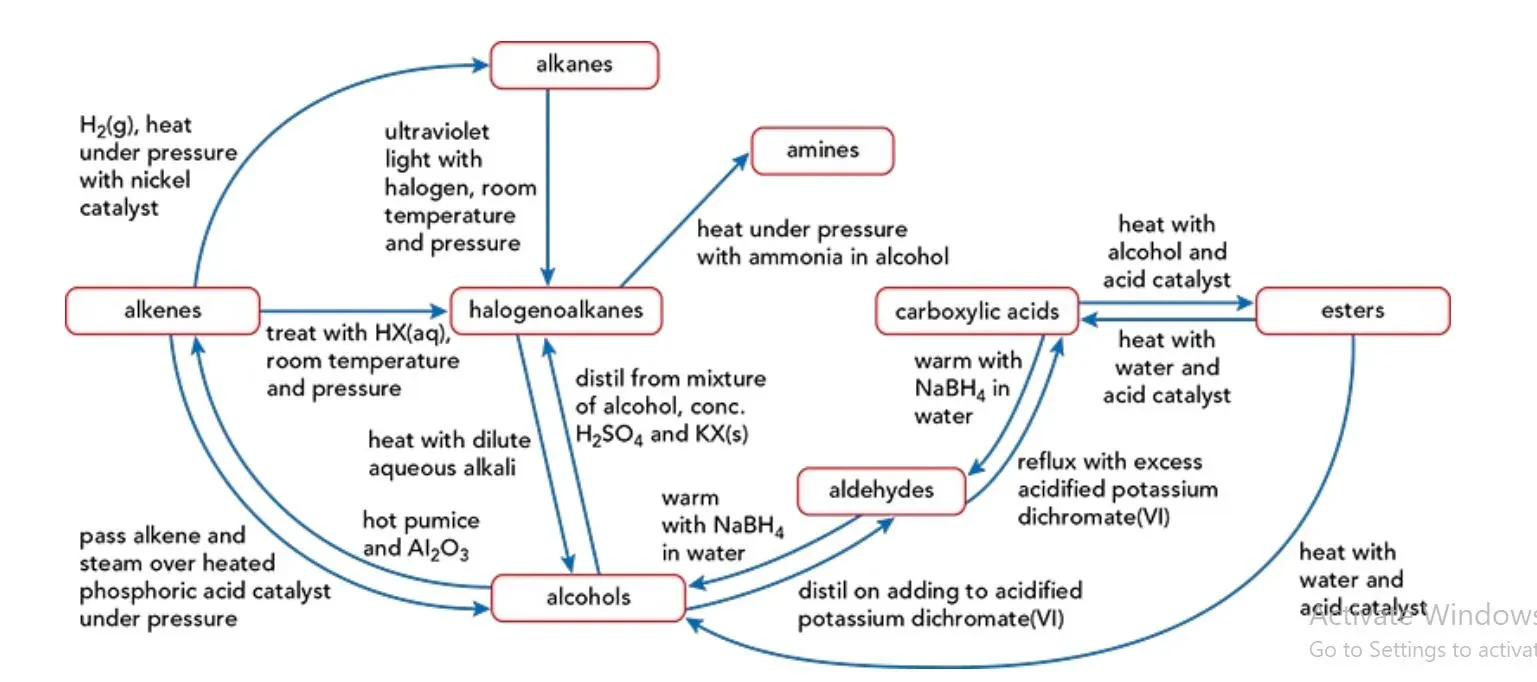
21.1.1 Adding Carbon Atoms
Sometimes, a starting material might not possess enough carbon atoms for the desired product. Carbon atoms can be effectively added to a molecule through specific reactions.
A. Nitrile Addition
The nitrile functional group, -CN, can be introduced to extend the carbon chain.
-
Formation of Nitrile from Alkyl Halide: Alkyl halides can be converted to nitriles by heating them with ethanolic potassium cyanide (KCN). Here, RCN contains one more carbon atom than the original RBr.
-
Further Transformations of Nitriles:
- Hydrolysis to Carboxylic Acid: Nitrile groups can be hydrolyzed to carboxylic acids by refluxing with dilute hydrochloric acid.
- Reduction to Amine: Nitriles can be reduced to primary amines by adding lithium aluminium hydride () in dry ether.
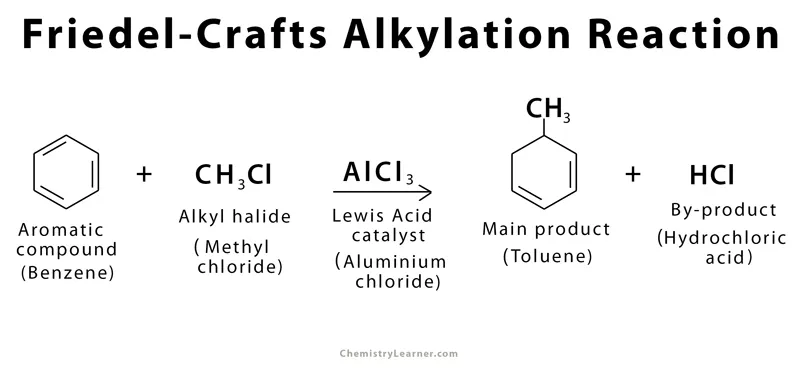
B. Friedel-Crafts Reaction
Friedel-Crafts reactions are important for adding alkyl or acyl side-chains to a benzene ring, thus increasing the carbon count of the aromatic system. For example, the alkylation of benzene:
21.1.2 Multi-step Synthetic Routes of Organic Molecules
Organic compounds are often synthesized via multi-step routes involving several functional group transformations.
Example: Preparation of Ethanoic Acid from Methane
This conversion involves three distinct steps:
-
Step 1: Methane to Chloromethane
- Type of reaction: Free Radical Substitution.
- Reagents: Chlorine gas () in the presence of UV light.
- By-product: Hydrochloric acid (HCl).
-
Step 2: Chloromethane to Cyanomethane
- Type of reaction: Nucleophilic Substitution.
- Reagents: Potassium cyanide (KCN).
-
Step 3: Cyanomethane to Ethanoic Acid
- Type of reaction: Hydrolysis.
- Reagents: Water () in the presence of acid (HCl).
21.1.3 Tests Used for the Identification of Functional Groups
Various chemical tests are employed to identify specific functional groups within an organic molecule.
Test 1: Bromine Water Test
- Purpose: Identification of unsaturation ( or ) in an organic compound.
- Reagent: Aqueous Bromine ().
- Observation with alkene or alkyne: The reddish-brown colour of bromine water fades or disappears, as bromine adds across the multiple bond.
Test 2: Silver Nitrate Test
- Purpose: Identification of halogen atoms in alkyl halides.
- Reagent: Aqueous silver nitrate () in ethanol.
- Observation with Chloroalkane (R-Cl): A white precipitate of silver chloride (AgCl) is formed.
- Observation with Bromoalkane (R-Br): A cream precipitate of silver bromide (AgBr) is formed.
- Observation with Iodoalkane (R-I): A yellow precipitate of silver iodide (AgI) is formed. (Note: The ethanol promotes the nucleophilic substitution of the halogen by the alcohol, forming an ether and a hydrogen halide, which then reacts with silver nitrate to form the silver halide precipitate.)
Test 3: Iodoform Test (Haloform Reaction)
- Purpose: Identification of methyl ketones () or methyl secondary alcohols (), and ethanol.
- Reagent: Iodine () and aqueous sodium hydroxide ().
- Conditions for positive test:
- Primary alcohol: Only ethanol ().
- Secondary alcohol: Only those where the -OH group is attached to carbon number 2, e.g., .
- Tertiary alcohol: Do not give a positive test.
- Aldehyde: Only ethanal ().
- Ketone: Only those where the carbonyl group () is at position 2, e.g., .
- Observation: A yellow precipitate of triiodomethane () is formed.
Test 4: 2,4-DNPH Test (Brady's Test)
- Purpose: Identifying test for carbonyl compounds (aldehydes and ketones).
- Reagent: 2,4-dinitrophenylhydrazine (2,4-DNPH).
- Observation: A yellow or orange precipitate of a dinitrophenylhydrazone derivative is formed.
Test 5: Fehling's Solution Test
- Purpose: Differentiating aldehydes from ketones (aldehydes give a positive test).
- Reagent: Fehling's solution (an alkaline solution of ions and sodium/potassium tartarate, acting as a mild oxidizing agent).
- Observation with aldehyde: A red precipitate of copper(I) oxide () is formed. Ketones give no reaction.
Test 6: Silver Mirror Test / Tollens' Test
- Purpose: Differentiating aldehydes from ketones (aldehydes give a positive test).
- Reagent: Ammoniacal silver nitrate (Tollens' reagent), a mild oxidizing agent.
- Observation with aldehyde: A silver mirror of elemental silver (Ag) is formed on the inner surface of the test tube. Ketones give no reaction.
Test 7: Distinguishing Primary, Secondary, and Tertiary Alcohols
- Purpose: To differentiate between primary, secondary, and tertiary alcohols based on their oxidation reactivity.
- Reagent: Potassium dichromate () in the presence of concentrated sulfuric acid ().
- Observation with Primary Alcohol (): The orange solution turns green. Primary alcohols are oxidized to aldehydes, which can be further oxidized to carboxylic acids (especially if heated or with excess oxidizing agent). To prepare only the aldehyde, the mixture is heated gently and the aldehyde is distilled off as it forms, preventing further oxidation.
- Observation with Secondary Alcohol (): The orange solution turns green. Secondary alcohols are oxidized to ketones. Ketones resist further oxidation under these conditions.
- Observation with Tertiary Alcohol (): No colour change of potassium dichromate (remains orange). Tertiary alcohols are generally resistant to oxidation under these conditions.
Test 8: Test of Carboxylic Acid
- Purpose: Identification of carboxylic acid.
- Reagent: Sodium carbonate () or sodium bicarbonate ().
- Observation: Effervescence (fizzing) due to the evolution of carbon dioxide gas ().
21.1.4 Types of Reaction Given by Functional Groups
1. Reactions of Alkanes
- Free Radical Substitution: Alkanes typically undergo free radical substitution reactions where a hydrogen atom is displaced by a free radical (e.g., a halogen radical).
- General reaction: Alkane + Chloroalkane + HCl
- Condition: Diffused sunlight (UV light).
2. Reactions of Unsaturated Hydrocarbons (Alkenes/Alkynes)
Unsaturated hydrocarbons, characterized by carbon-carbon double or triple bonds, predominantly undergo two main types of reactions:
-
i. Electrophilic Addition Reactions: In these reactions, one of the bonds in the carbon-carbon multiple bond (usually the -bond) is broken, and two new single bonds are formed from each of the two carbon atoms to the incoming electrophile. A single product is always formed.
-
ii. Oxidation Reactions:
- Diol Formation (Hydroxylation): Alkenes can be oxidized to produce diols (compounds with two hydroxyl groups) in the presence of a cold, dilute, alkaline solution of potassium permanganate (, Baeyer's test).
- Ozonolysis: Alkenes can undergo ozonolysis, which breaks the -bond and -bond, forming two new carbonyl compounds (aldehydes and/or ketones). Alkynes upon ozonolysis yield carboxylic acids.
3. Reactions of Alkyl Halides
Alkyl halides (R-X) are versatile compounds that undergo two main types of reactions:
-
i. Nucleophilic Substitution Reactions: The halogen group (-X) in an alkyl halide is displaced by an attacking nucleophile (e.g., , , ).
-
ii. Elimination Reactions: In an elimination reaction, a hydrogen atom and a halogen atom are removed from adjacent carbon atoms of an alkyl halide, resulting in the formation of an alkene.
- Reagent: Ethanolic sodium hydroxide () or potassium hydroxide ().
4. Reactions of Alcohols
Alcohols undergo a variety of reactions, including oxidation, substitution, dehydration, and esterification.
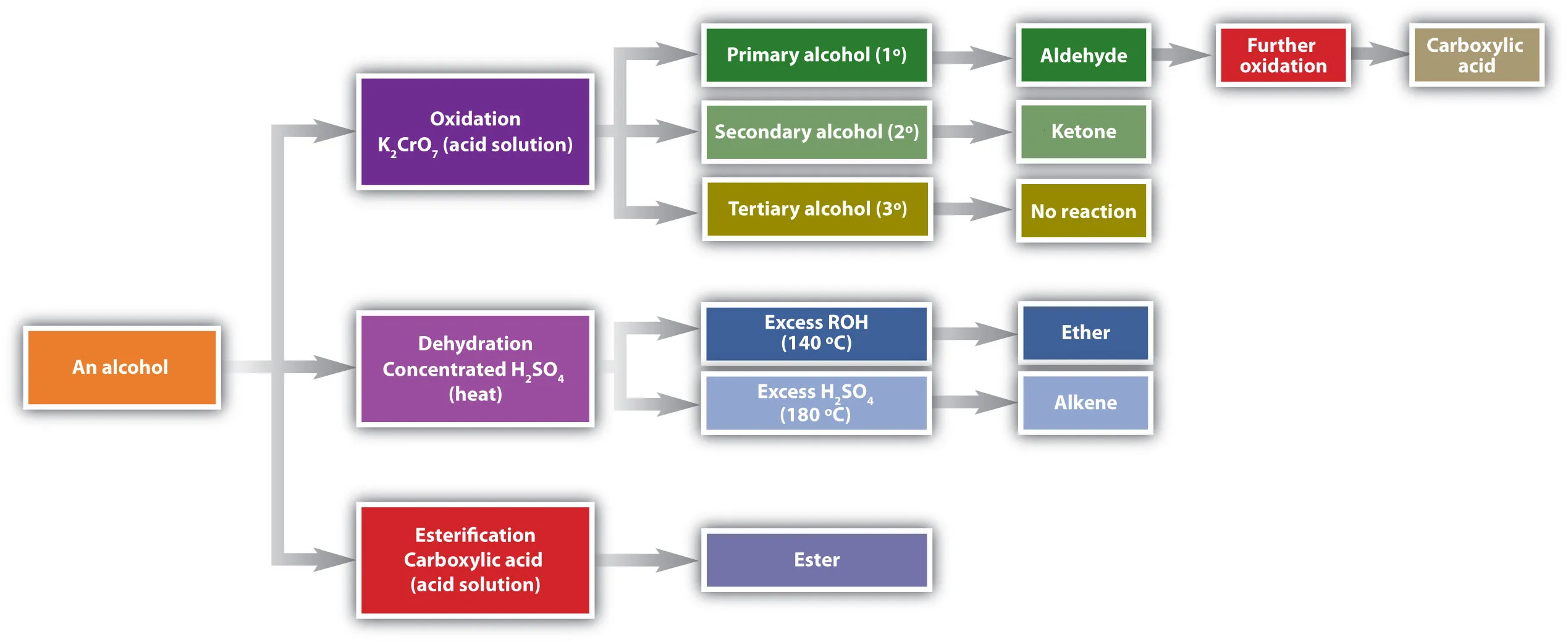
5. Reactions of Carbonyl Compounds (Aldehydes and Ketones)
Carbonyl compounds primarily engage in nucleophilic addition reactions, along with characteristic oxidation and reduction reactions.
- Nucleophilic Addition: The carbon-oxygen double bond is susceptible to attack by nucleophiles, often leading to the formation of hydroxyl nitriles (cyanohydrins).
- Oxidation: Aldehydes are readily oxidized to carboxylic acids, while ketones generally resist oxidation under mild conditions.
- Reduction: Aldehydes are reduced to primary alcohols, and ketones are reduced to secondary alcohols.
6. Reactions of Carboxylic Acids
Carboxylic acids exhibit acidic properties and undergo reactions characteristic of acids, as well as esterification.
- Acid-Base Reactions: Carboxylic acids react with bases, metals, and metal carbonates, liberating hydrogen gas (with active metals) or carbon dioxide (with carbonates).
- Esterification: The reaction of a carboxylic acid with an alcohol, typically in the presence of an acid catalyst, forms an ester.
Possible Questions/Answers
-
Q: What is the key characteristic of an electrophilic addition reaction in alkenes? A: The breaking of a -bond and the formation of two new -bonds, resulting in a single product.
-
Q: Why do tertiary alcohols not react with acidified potassium dichromate? A: Tertiary alcohols do not have a hydrogen atom directly attached to the carbon bearing the hydroxyl group, which is required for oxidation to occur under these conditions.
-
Q: What is the purpose of adding in a synthetic route starting from a carboxylic acid? A: To convert the carboxylic acid into a more reactive acyl chloride, which can then undergo further reactions (e.g., with amines to form amides).
-
Q: How would you distinguish between propanal and propanone using chemical tests? A: Use Fehling's solution or Tollens' reagent. Propanal (an aldehyde) will give a positive test (red ppt with Fehling's, silver mirror with Tollens'), while propanone (a ketone) will not react.