20.2 Physical Properties of Amines
Amines exhibit distinct physical properties primarily due to the nature of the nitrogen-hydrogen bond. Lower aliphatic amines like methylamine and ethylamine are gases at room temperature and possess a characteristic ammonia-like or "fishy" odor.
Polarity and Hydrogen Bonding
The bond between nitrogen and hydrogen () is polar because of the significant electronegativity difference between the two atoms. This polarity allows primary () and secondary () amine molecules to form hydrogen bonds with each other.
Boiling and Melting Points
Due to intermolecular hydrogen bonding, amines require more energy to change phase compared to nonpolar compounds of similar molar mass.
- They have higher melting and boiling points than analogous alkanes.
- Example: Ethanamine (, boiling point 17°C) has a much higher boiling point than propane (, boiling point -42°C), despite having a similar molar mass.
- Comparison between Amines: Primary amines generally have higher boiling points than secondary amines of the same molecular weight because the nitrogen in a primary amine has two hydrogens available for hydrogen bonding. Tertiary amines, having no bonds, cannot form intermolecular hydrogen bonds with each other and thus have the lowest boiling points among isomers.
Solubility
The ability to form hydrogen bonds also explains their solubility in water.
- Lower molecular weight amines (primary, secondary, and even tertiary) are soluble in water because they can form hydrogen bonds with water molecules.
- As the size of the alkyl group increases, the hydrophobic (nonpolar) character of the molecule dominates, and solubility in water decreases.
Structure of Amines
The geometry and electronic structure of the nitrogen atom are key to understanding the chemical behavior of amines.
Hybridization and Geometry
In amines, the central nitrogen atom is -hybridized. This results in a nearly tetrahedral geometry around the nitrogen. Nitrogen forms three sigma () bonds with its three -hybrid orbitals. The fourth -hybrid orbital is non-bonding and holds a lone pair of electrons. Because the lone pair exerts more repulsion than bonding pairs, the bond angles (e.g., ) are slightly less than the ideal .
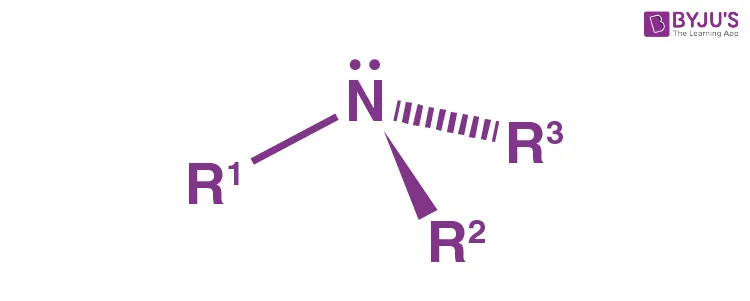
Basicity and Nucleophilicity
The lone pair of electrons on the nitrogen atom can be donated to an electron-deficient species (an electrophile or a proton). This ability to donate its electron pair makes an amine a Lewis base and a nucleophile.
Optical Activity
A tertiary amine with three different groups attached to the nitrogen atom is chiral (asymmetric). Chiral molecules are optically active, meaning they can rotate the plane of plane-polarized light. However, many chiral amines cannot be resolved into separate enantiomers at room temperature because of rapid pyramidal inversion, which interconverts the two enantiomers like an umbrella turning inside out.