17.2 Physical Properties of Alkyl Halides
This section explores the physical characteristics of alkyl halides (halogenoalkanes), focusing on their melting points, boiling points, and the underlying structural reasons for these properties.
Melting and Boiling Points
Alkyl halides generally have higher melting and boiling points compared to alkanes with a similar number of carbon atoms.
Reason: The primary reason is the polarity of the carbon-halogen (C–X) bond. Since halogens (F, Cl, Br, I) are more electronegative than carbon, the C–X bond is polar covalent. This polarity creates permanent dipole-dipole interactions between alkyl halide molecules, which are stronger than the weak London dispersion forces present in nonpolar alkanes. Overcoming these stronger intermolecular forces requires more energy, resulting in higher boiling and melting points.
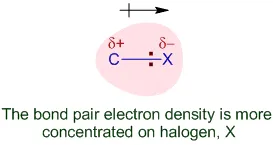
Structure of Alkyl Halides
The molecular structure of alkyl halides dictates their physical and chemical properties.
- Hybridization: In an alkyl halide (), the carbon atom bonded to the halogen is hybridized. This results in a tetrahedral geometry around that carbon atom.
- Bonding: The carbon and halogen atoms are joined by a strong sigma () bond.
- Polarity: The significant difference in electronegativity between carbon and the halogen atom makes the C–X bond polar. The electron density is more concentrated on the halogen atom (), leaving the carbon with a partial positive charge ().
Electrophilic Nature of the Carbon in C–X Bond
The polarity of the C–X bond has an important consequence for reactivity:
- The carbon atom bonded to the halogen carries a partial positive charge (), making it an electrophilic centre.
- This electron-deficient carbon is susceptible to attack by nucleophiles (electron-pair donors).
- This is the fundamental reason why alkyl halides undergo nucleophilic substitution and elimination reactions.
Summary
| Property | Alkyl Halides vs. Alkanes |
|---|---|
| Boiling/Melting Point | Higher (due to dipole-dipole forces) |
| C–X Bond Type | Polar covalent bond |
| Carbon Hybridization | (tetrahedral) |
| Carbon Character | Electrophilic () |
- Alkyl halides possess higher melting and boiling points than alkanes of comparable size.
- This is attributed to the polarity of the C–X bond, which leads to stronger dipole-dipole intermolecular forces.
- The carbon atom in the C–X bond is hybridized and forms a bond with the halogen.
- The polarity of the C–X bond makes the carbon atom electrophilic, which is central to the reactivity of alkyl halides in substitution and elimination reactions.