17.1 Types of Halogenoalkanes
17.1 Types of Halogenoalkanes
Halogenoalkanes (or alkyl halides) are organic compounds containing one or more halogen atoms (F, Cl, Br, I) bonded to an alkyl group. They are classified based on the number of halogen atoms and the type of carbon atom to which the halogen is attached.
Classification by Number of Halogen Atoms
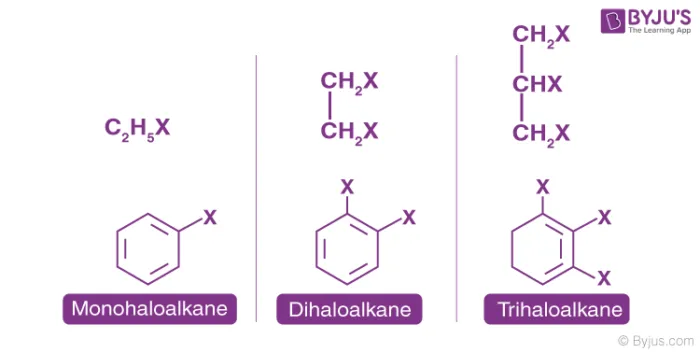
Halogenoalkanes can be classified as mono, di, tri, or poly haloalkanes depending on how many halogen atoms are present in the molecule.
- Monohaloalkane (One halogen atom): General formula .
- Dihaloalkane (Two halogen atoms): These can be gem-dihalides (both halogens on the same carbon) or vic-dihalides (halogens on adjacent carbons).
- Trihaloalkane/Polyhaloalkane (Three or more halogen atoms)
Classification by Position of Halogen Atom
Halogenoalkanes are also classified into primary (1°), secondary (2°), and tertiary (3°) types. This classification depends on the nature of the carbon atom to which the halogen is directly attached.
1. Primary (1°) Alkyl Halides
"Alkyl halide in which halogen atom is attached with a primary carbon are called primary alkyl halides."
A primary carbon is a carbon atom that is bonded to only one other carbon atom (or no other carbon atoms, as in methyl halides).
Examples:
- Methyl chloride (Chloromethane):
- Ethyl chloride (Chloroethane):
- n-Propyl chloride (1-Chloropropane):
In these examples, the halogen is attached to a primary () carbon atom.
2. Secondary (2°) Alkyl Halides
"Alkyl halide in which halogen atom is attached with a secondary carbon atom is called secondary alkyl halide."
A secondary carbon is a carbon atom that is bonded to two other carbon atoms.
Examples:
- 2-Chloropropane (Isopropyl chloride):
- 2-Chlorobutane (sec-Butyl chloride):
3. Tertiary (3°) Alkyl Halides
"Alkyl halides, in which halogen atom is attached to a tertiary carbon is called tertiary alkyl halide."
A tertiary carbon is a carbon atom that is bonded to three other carbon atoms.
Example:
- 2-Methyl-2-chloropropane (t-Butyl chloride):
| Class | Description | General Structure (R = alkyl, X = halogen) | Example |
|---|---|---|---|
| Primary (1°) | Halogen is bonded to a carbon attached to one or zero other carbon atoms. | or | Chloroethane () |
| Secondary (2°) | Halogen is bonded to a carbon attached to two other carbon atoms. | 2-Chloropropane () | |
| Tertiary (3°) | Halogen is bonded to a carbon attached to three other carbon atoms. | 2-Methyl-2-chloropropane () |
References
(Derived from FBISE textbook)
12.1 Halogens→(/chemistry-11/12-halogens/12-1-halogens)