16.5 Molecular Structure of Ethene
Ethene () is an alkene consisting of two carbon atoms joined by a double covalent bond, with two hydrogen atoms singly bonded to each carbon.
- Hybridization: Each carbon atom in ethene is hybridized.
- Geometry: The hybridization results in a trigonal planar arrangement around each carbon atom.
- Planarity: The entire ethene molecule is planar, meaning all six atoms (2 carbons and 4 hydrogens) lie in the same plane.
- Bond Angles: The bond angle is approximately and the bond angle is approximately (both close to the ideal ).
- Bond Length: The bond length is , shorter than the single bond () in ethane.
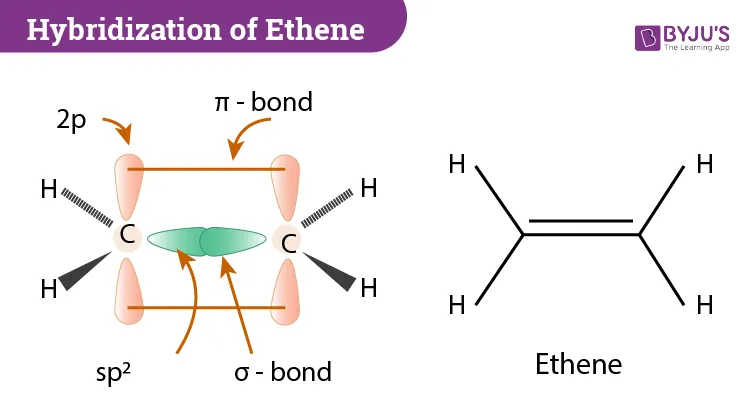
Bonding in Ethene
The double bond between the two carbon atoms is a combination of one sigma () bond and one pi () bond.
Sigma () Bonds
Sigma bonds are formed by the direct, head-on overlap of atomic or hybrid orbitals. They are strong and hold electrons tightly.
- Carbon-Carbon () Bond: Formed by the head-on overlap of two hybridized orbitals, one from each carbon atom ( overlap).
- Carbon-Hydrogen () Bonds: Each of the four C-H bonds is formed by the overlap of an orbital from a carbon atom and the -orbital of a hydrogen atom ( overlap).
Pi () Bond
Pi bonds are formed by the parallel (sideways) overlap of unhybridized p-orbitals. For more on orbital shapes, see Shapes of Orbitals→.
- Carbon-Carbon () Bond: After hybridization, each carbon atom has one unhybridized -orbital perpendicular to the molecular plane. These two parallel -orbitals overlap sideways to form the bond.
- Characteristics: The electrons in a bond are held less tightly and are located above and below the plane of the sigma bonds. This makes the bond weaker and more exposed than a sigma bond.
Why is the C=C Bond Shorter than C-C?
Two factors contribute to the shorter bond () compared to ():
- Greater -character: orbitals have 33% -character vs. 25% in orbitals, making them more compact and pulling the nuclei closer.
- Additional bond: The extra electron density between the nuclei from the bond pulls them closer together.
Reactivity of Ethene
The presence of the weak and accessible bond makes ethene significantly more reactive than alkanes like ethane.
Ethene vs. Ethane
| Property | Ethene () | Ethane () |
|---|---|---|
| Bond type | + | only |
| electrons | Exposed, accessible | None |
| Typical reaction | Electrophilic addition | Free radical substitution |
| Reactivity | Higher | Lower |
Ethene is more reactive than ethane because the bond can be easily broken to form new single bonds (addition reactions). Ethane only contains strong bonds, which are difficult to break.
Ethene vs. Ethyne
Ethene is more reactive than ethyne. The electrons in ethene are more exposed and accessible for reaction. The shorter triple bond in ethyne holds its electrons more tightly between the carbon nuclei (due to greater nuclear attraction), making them less available for electrophilic attack compared to the more diffuse cloud of the double bond in ethene.
Electrophilic Addition
Alkenes typically undergo electrophilic addition reactions:
- The exposed electrons act as a nucleophile (electron donor)
- An electrophile (electron acceptor) is attracted to the cloud
- The bond breaks and two new bonds form
Example: Addition of to ethene: