11.1 Nitrogen
Nitrogen, a nonmetallic element in Group 15 (formerly Group V) of the periodic table, constitutes approximately 78% of the Earth's atmosphere. It primarily exists as diatomic molecules (). Nitrogen gas is known for its low reactivity.
Electronic Configuration and Bonding
To understand the inert nature of nitrogen, we must examine its molecular bonding.
- The electronic configuration of a nitrogen atom is .
Rules of Electronic Configuration→
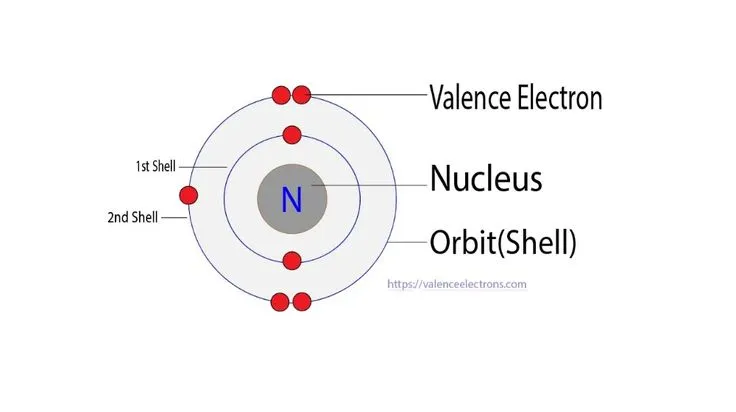
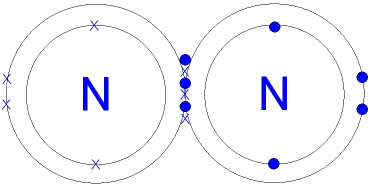
- Each nitrogen atom needs to gain three electrons to achieve a stable noble gas configuration (like Neon).
- Two nitrogen atoms accomplish this by sharing three pairs of electrons, forming a triple covalent bond ().
- This triple bond is extremely strong, with a bond energy of nearly (often rounded to ). The large amount of energy required to break this bond is the reason for the chemical inertness of nitrogen gas (). Consequently, nitrogen only reacts under extreme conditions of temperature and pressure.
Ammonia and the Ammonium Ion
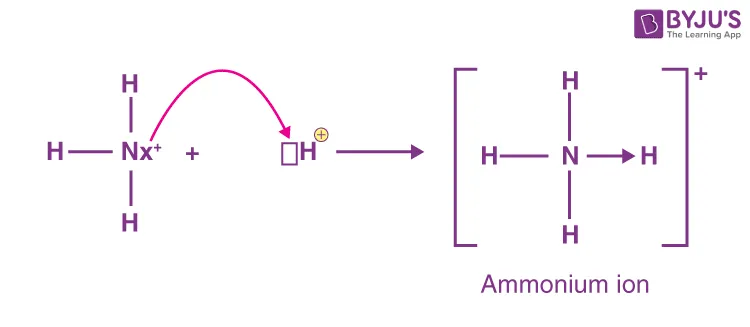
Formation of the Ammonium Ion ()
The ammonium ion is formed when ammonia (), an alkaline gas, reacts with an acid. Ammonia's alkaline nature is evident as it turns moist red litmus paper blue.
The reaction with a hydrogen ion () is:
Mechanism (Lewis Acid-Base Theory):
- The nitrogen atom in ammonia has a lone pair of electrons.
- It donates this lone pair to a hydrogen ion () to form a dative (or coordinate) covalent bond.
- According to the Lewis concept, an electron-pair donor is a base. This is why ammonia is basic.
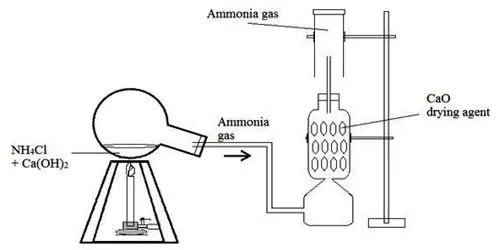
Laboratory Preparation of Ammonia Gas
In the laboratory, ammonia gas is prepared by heating an ammonium salt with a base.
- Reactants: Ammonium chloride () and calcium hydroxide (), both solids.
- Word Equation: Ammonium Chloride + Calcium Hydroxide Calcium Chloride + Ammonia + Water
- Chemical Equation:
The Haber Process: Industrial Manufacture of Ammonia
Ammonia is manufactured on a large scale via the Haber Process, which involves the direct combination of nitrogen and hydrogen gases.
-
Main Equation:
-
Conditions for the Haber Process:
- Temperature:
- Pressure: 200 atm
- Catalyst: Iron (Fe) with promoters and
-
Explanation of Conditions:
- The forward reaction is exothermic. According to Le Chatelier's principle, a lower temperature would favor a higher yield of ammonia. However, the reaction rate would be too slow. Therefore, a compromise temperature is used to achieve a reasonable rate and yield.
- The forward reaction involves a decrease in the number of moles of gas (4 moles on the left, 2 moles on the right). High pressure shifts the equilibrium to the right, favoring the production of ammonia.
- The iron catalyst lowers the activation energy, allowing the reaction to proceed at a practical rate at the compromise temperature. It does not affect the equilibrium position.
Oxides of Nitrogen ()
Nitrogen forms several oxides:
| Oxide | Formula | Common Name |
|---|---|---|
| Dinitrogen monoxide | Laughing gas | |
| Nitrogen monoxide | Nitric oxide | |
| Nitrogen dioxide | — |
Formation of Nitrogen Oxides
-
Natural Formation (Thunderstorms): The high energy from lightning provides the activation energy for nitrogen and oxygen in the air to react, forming nitrogen monoxide.
-
Internal Combustion Engines: The high temperature and pressure inside an engine also cause nitrogen and oxygen from the air to react, producing .
-
Oxidation to Nitrogen Dioxide: The nitrogen monoxide formed is quickly oxidized by oxygen in the air to form brown nitrogen dioxide.
Acid Rain Formation
Nitrogen dioxide dissolves in water droplets in the atmosphere to form nitric acid, which then falls as acid rain.
Nitric acid in the soil is converted to nitrates, which are essential fertilizers for plant growth and protein synthesis. However, excess acid rain damages ecosystems and corrodes structures.
Catalytic Removal of Nitrogen Oxides from Exhaust Gases
To reduce air pollution, vehicles are equipped with catalytic converters to remove harmful gases like nitrogen monoxide () from the exhaust.
-
Device: A three-way catalytic converter.
-
Catalysts: A mixture of Platinum (Pt), Palladium (Pd), and Rhodium (Rh).
-
Function: The converter facilitates redox reactions that convert:
- → (reduction)
- Unburned hydrocarbons and → and (oxidation)
-
Overall effect: Harmful exhaust gases are converted into less harmful , , and before being released into the atmosphere.
Example reaction: