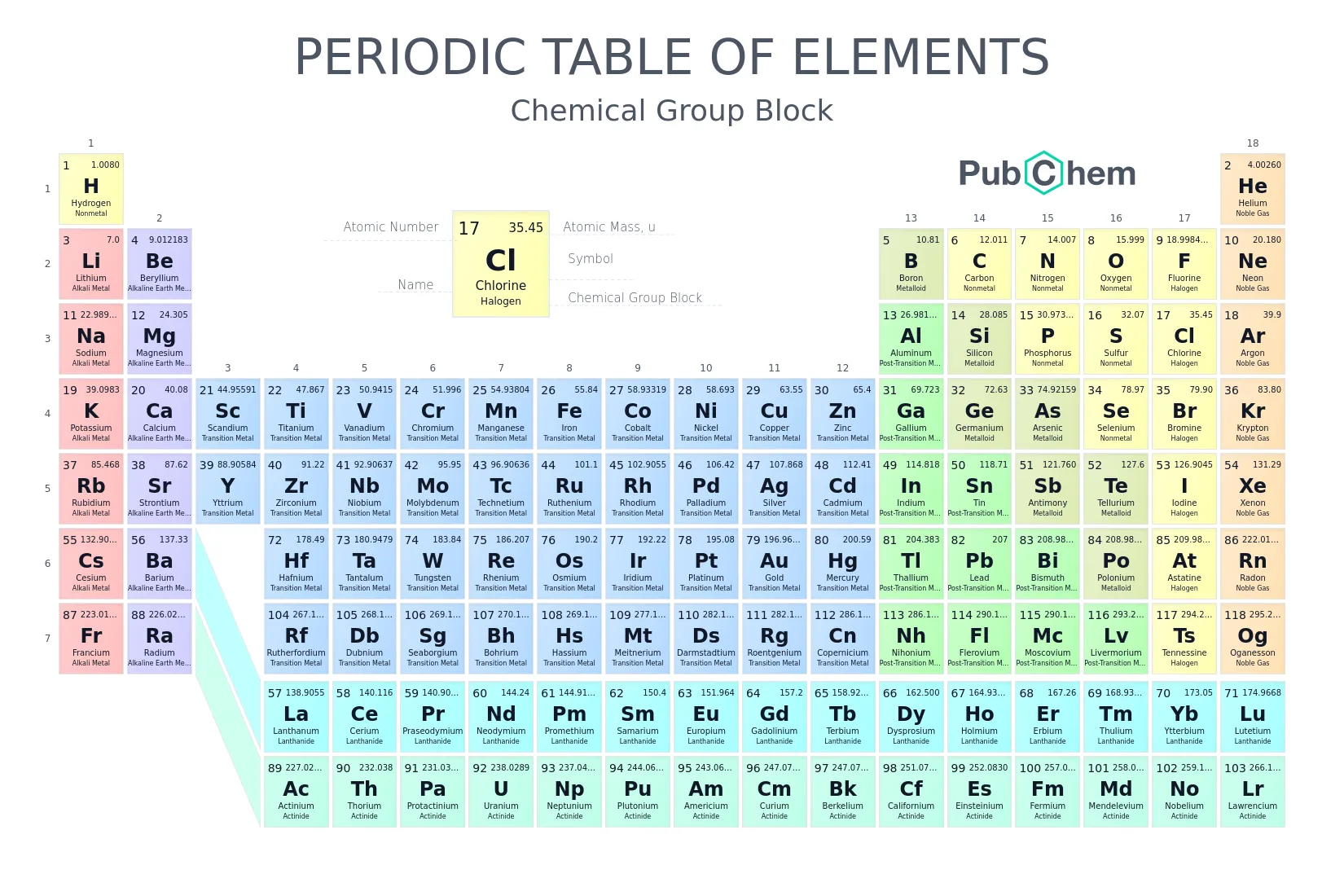
10.1 Periods and Groups of Elements
The periodic table is a systematic arrangement of elements based on the periodic law. It organizes elements into horizontal rows (periods) and vertical columns (groups) according to their increasing atomic number and recurring chemical properties. The position of an element is determined by its electronic configuration.
Rules of Electronic Configuration→
Periods
-
Definition: The seven horizontal rows in the periodic table are called periods.
-
Arrangement: Elements are listed in order of ascending atomic number from left to right.
-
Number of Elements: The number of elements in each period varies based on the filling of shells.
-
Period 1: 2 elements (Hydrogen, and Helium, )
-
Periods 2 and 3: 8 elements each
-
Periods 4 and 5: 18 elements each
-
Periods 6 and 7: 32 elements each
-
Types of Periods:
- Short Periods: Periods 1, 2, and 3.
- Long Periods: Periods 4, 5, 6, and 7.
-
Property Trends: The properties of elements change gradually and predictably across a period. This pattern of properties repeats in the next period, which is the essence of the periodic law.
Groups
-
Definition: The vertical columns of elements in the periodic table are called groups or families. Elements within the same group share similar chemical properties because they have the same outer electronic configuration.
-
Group Numbering Systems:
-
Traditional (Old IUPAC): Uses Roman numerals and letters (A and B).
- Group A: Representative or main group elements (Groups IA to VIIA).
- Group B: Transition elements.
-
Modern IUPAC (1988): Numbers the groups 1 through 18 from left to right.
-
-
Valence Electrons and Group Number:
For main group elements (Groups 1, 2, and 13-18), the group number indicates the number of valence electrons (electrons in the outermost energy level).
- Group 1 (IA): 1 valence electron
- Group 2 (IIA): 2 valence electrons
- Group 13 (IIIA): 3 valence electrons (Group Number 10)
- Group 14 (IVA): 4 valence electrons (Group Number 10)
- Group 15 (VA): 5 valence electrons (Group Number 10)
- Group 16 (VIA): 6 valence electrons (Group Number 10)
- Group 17 (VIIA): 7 valence electrons (Group Number 10)
- Group 18 (VIIIA): 8 valence electrons (except Helium, which has 2)
Blocks of Elements
Elements are also classified into blocks based on the subshell that is in the process of being filled.
- s-block: Groups 1 and 2 (Alkali and Alkaline Earth metals).
- p-block: Groups 13 to 18.
- d-block: Transition elements (Groups 3 to 12).
- f-block: Lanthanides and Actinides (placed at the bottom).
Special Group Names
Certain groups of elements have common names based on their properties.
| Group Number (Modern) | Group Number (Traditional) | Special Name | Description |
|---|---|---|---|
| 1 | IA | Alkali Metals | Highly reactive metals |
| 2 | IIA | Alkaline Earth Metals | Reactive metals, but less so than alkali metals |
| 13 | IIIA | Boron Family | Contains one metalloid and several metals |
| 16 | VIA | Chalcogens / Oxygen Family | Includes nonmetals, metalloids, and a metal |
| 17 | VIIA | Halogens | Highly reactive nonmetals |
| 18 | VIIIA | Noble Gases | Very unreactive (inert) gases |