3.5 Role of Respiratory Pigments
Respiratory pigments are coloured, protein-based molecules that act as oxygen carriers by binding reversibly to oxygen. They all contain a coloured, non-protein component, such as the haem group in haemoglobin and myoglobin. These pigments are essential for meeting the high oxygen demands of aerobic respiration in large, active multicellular organisms.
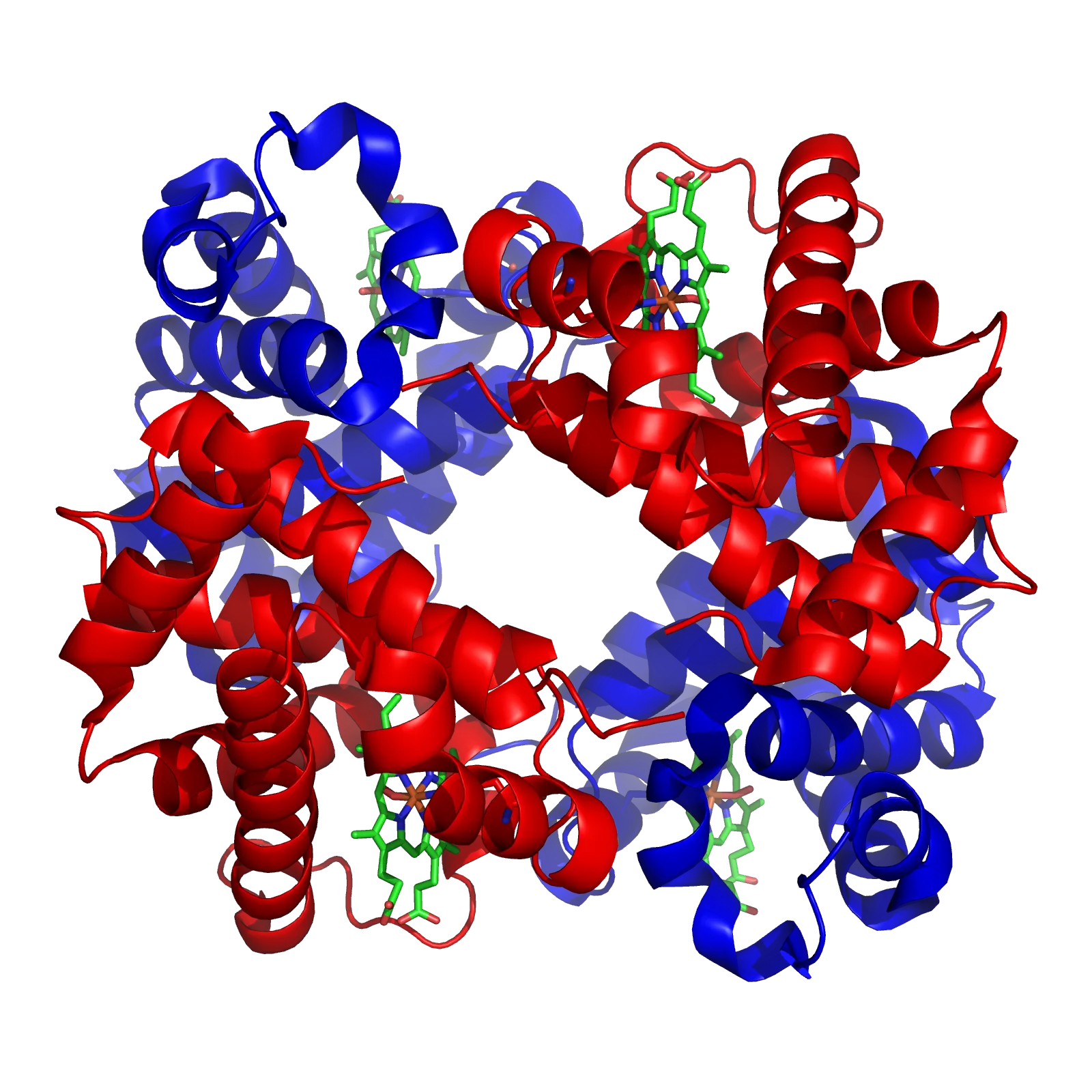
Haemoglobin (Hb)
Structure: Composed of four globin protein chains. Each chain is associated with a haem group, which is an iron-containing ring structure.
Function: The iron atom in each haem group combines loosely and reversibly with one oxygen molecule. Therefore, one haemoglobin molecule can carry up to four oxygen molecules.
Oxygen Binding: It binds to oxygen in environments with high oxygen concentration (like the lungs) and releases oxygen in environments with low oxygen concentration (like respiring tissues). This process is influenced by cooperativity, where the binding of one oxygen molecule makes it easier for subsequent molecules to bind.
Location: Found in Red Blood Cells (RBCs). At normal arterial (100 mmHg), 100ml of blood carries approximately 20ml of oxygen.
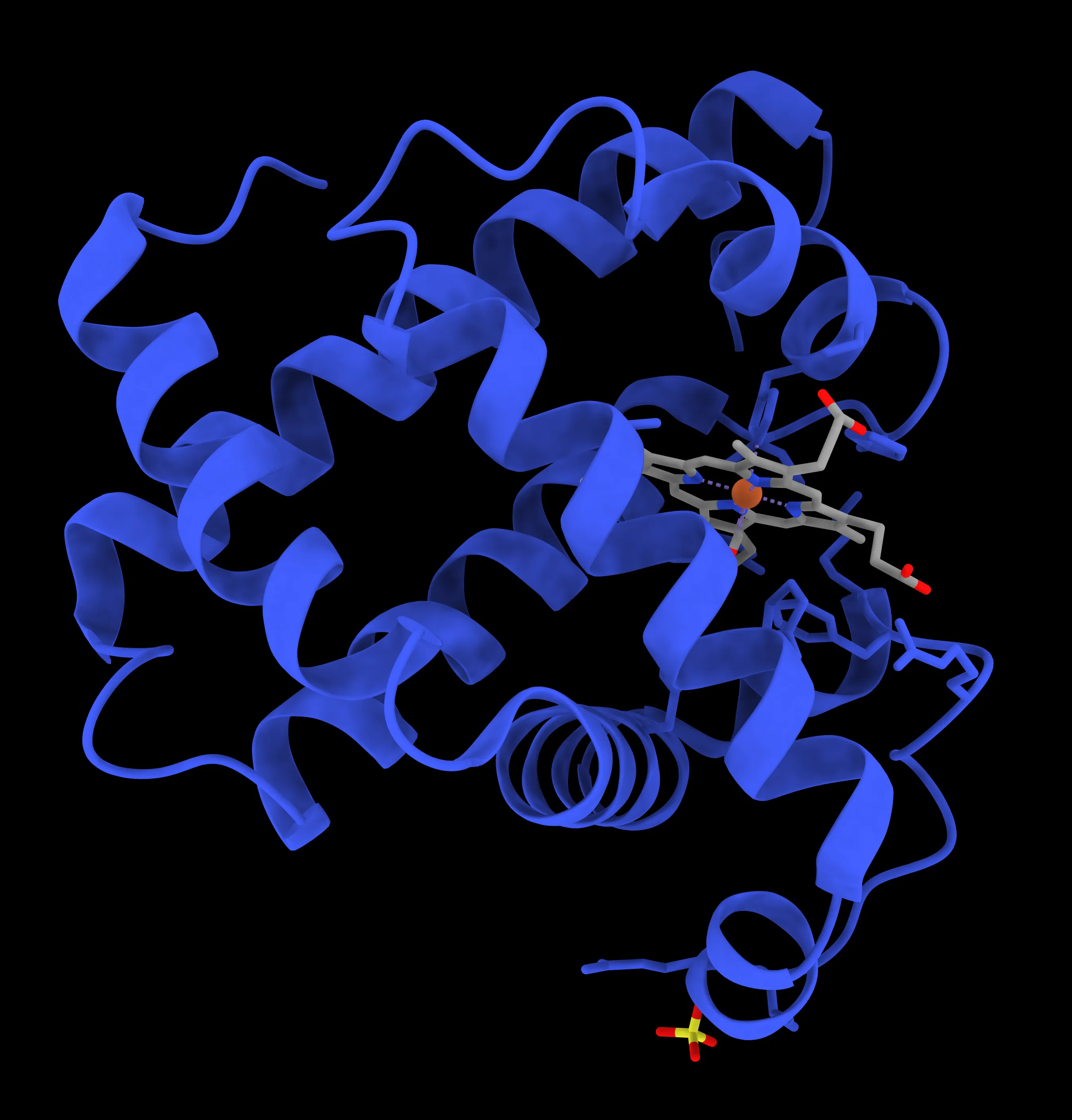
Myoglobin (Mb)
Structure: Consists of a single polypeptide chain associated with one iron-containing haem group. It can bind to only one molecule of oxygen.
Function: Serves as an oxygen storage molecule and an intermediate for transferring oxygen from haemoglobin to the aerobic metabolic processes within muscle cells.
Oxygen Binding: It has a higher affinity for oxygen than haemoglobin. It only releases its stored oxygen when the partial pressure of oxygen () drops below 20 mmHg, such as during strenuous exercise when oxyhaemoglobin supplies are depleted.
Location: Found in skeletal muscles, giving meat its red colour.
Comparison: Haemoglobin vs. Myoglobin
| Feature | Haemoglobin | Myoglobin |
|---|---|---|
| Structure | Four polypeptide chains | One polypeptide chain |
| Haem Groups | Four per molecule | One per molecule |
| Capacity | Binds up to four molecules | Binds one molecule |
| Location | Red Blood Cells (RBCs) | Muscle tissue |
| Primary Function | Oxygen transport | Oxygen storage |
| Oxygen Affinity | Lower affinity | Higher affinity |
| Release | Loses at ≈ 60 mmHg | Loses at < 20 mmHg |
Factors Affecting Oxygen Binding (Bohr Effect)
The affinity of haemoglobin for oxygen is affected by environmental factors. The Bohr Effect describes how an increase in concentration or a decrease in pH (acidic conditions) decreases haemoglobin's affinity for oxygen, causing the dissociation curve to shift to the right. This facilitates the release of oxygen to active tissues.
Carbon Monoxide (CO) Poisoning
Cause: Inhaling carbon monoxide, often produced by incomplete combustion from sources like faulty gas heaters in enclosed spaces.
Mechanism:
- CO binds to the iron atom in haemoglobin with an affinity ~250 times greater than that of oxygen.
- This binding forms a stable compound called carboxyhaemoglobin, which prevents haemoglobin from picking up oxygen in the lungs.
- CO also increases haemoglobin's affinity for any oxygen it is still carrying, preventing it from releasing that oxygen to the tissues.
Symptoms: Nausea, vomiting, headache, changes in mental status, and a characteristic cherry-red lip colour.