3.4 Transport of Gases
The transport of respiratory gases, oxygen () and carbon dioxide (), is a critical function of the circulatory system, facilitated by the blood. Blood carries from the lungs to the body's tissues and transports from the tissues back to the lungs for exhalation.
3.4.1 Transport of Oxygen in Blood
Oxygen is transported from the lungs to the tissues in two main ways:
- As Oxyhaemoglobin (97%): The vast majority of binds reversibly with haemoglobin (Hb), a protein found in red blood cells (RBCs), to form oxyhaemoglobin ().
- Dissolved in Plasma (3%): A small fraction of dissolves directly into the blood plasma.
Formation of Oxyhaemoglobin
- In the lungs, where the partial pressure of oxygen () is high, readily binds to the iron-containing heme groups of haemoglobin.
- This reaction is reversible.
- Each haemoglobin molecule can bind with up to four oxygen molecules.
Oxygen-Carrying Capacity of Blood
- This is defined as the maximum amount of that can be transported by the blood.
- It is directly proportional to the partial pressure of oxygen ().
- Factors affecting binding:
- High (in lungs) promotes binding.
- Low , low pH (high ), high , and high temperature (in tissues) promote the release of from haemoglobin. This phenomenon where high and low pH decrease hemoglobin's affinity for oxygen is known as the Bohr Effect.
| Blood Type | (mmHg) | Content (ml/100ml) | Hb Saturation |
|---|---|---|---|
| Arterial Blood | 95 | 19.4 | 97% |
| Venous Blood | 40 | 14.4 | 75% |
- Note: The maximum capacity is 20 ml / 100 ml of blood (100% saturation at 100 mmHg). On average, 5 ml of is delivered to the tissues by every 100 ml of blood under normal conditions.
3.4.2 Transport of Carbon Dioxide in Blood
Carbon dioxide, a waste product of cellular metabolism, is transported from the tissues to the lungs in three forms:
| Method of Transport | Percentage |
|---|---|
| Bicarbonate ions () | ~70% |
| Carbaminohaemoglobin | ~23% |
| Dissolved in Plasma | ~7% |
(I) As Bicarbonate Ions ()
This is the primary method for transport.
- Diffusion: diffuses from tissue cells into the blood and enters Red Blood Cells (RBCs).
- Carbonic Acid Formation: Inside the RBC, rapidly combines with water () to form carbonic acid (). This reaction is catalyzed by the enzyme carbonic anhydrase.
- Dissociation: Carbonic acid is unstable and quickly dissociates into hydrogen ions () and bicarbonate ions ().
- Buffering: The released ions are buffered by haemoglobin, which binds them to form haemoglobinic acid (HHb). This process also facilitates the release of oxygen to the tissues (Bohr effect).
-
Chloride Shift (Hamburger's Phenomenon): To maintain electrical neutrality, as bicarbonate ions () move out of the RBC into the plasma, chloride ions () move into the RBC from the plasma.
-
In the Lungs: The entire process is reversed. re-enters the RBC, combines with to form carbonic acid, which then breaks down into and . The diffuses into the alveoli to be exhaled.
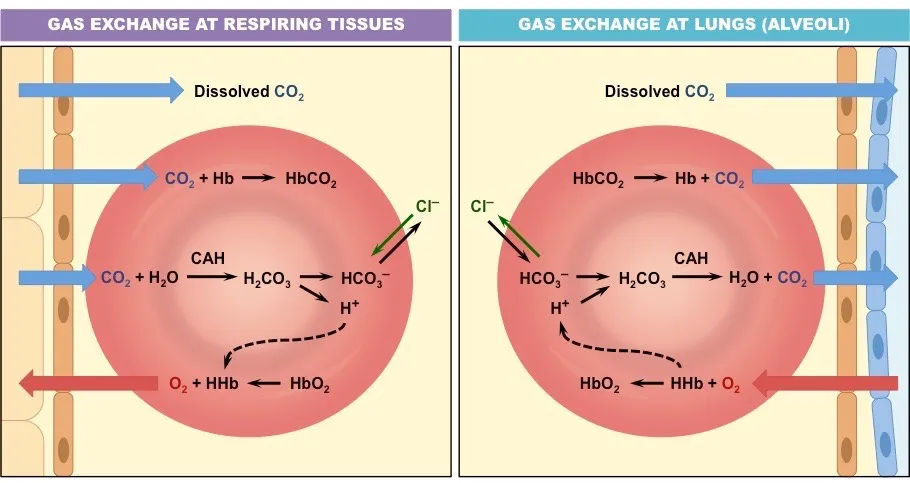
(II) As Carbaminohaemoglobin
- binds directly to the globin (protein) part of haemoglobin, not the heme group.
- This binding is reversible and dependent on the partial pressure of ().
- Formation occurs in the tissues where is high.
- Dissociation occurs in the lungs where is low.
(III) As Dissolved in Plasma
- A small amount of dissolves directly in the blood plasma. This is the least efficient transport mechanism.