2.2 Different Types of Bonds and Interactions in Biological Molecules
The structural stability and function of biological molecules depend on a variety of chemical bonds and weaker interactions. These forces can occur within a single molecule (intramolecular) or between different molecules (intermolecular).
Chemical Bonds
These are strong forces that hold atoms together within a molecule.
Covalent Bonds
Definition: Formed by the mutual sharing of electrons between two atoms to achieve a stable electron configuration.
Significance: As biomolecules are primarily composed of non-metals (C, H, O, N, S, P), covalent bonds are the most prevalent and fundamental type, forming the backbones of macromolecules.
Examples in Biomolecules:
- Peptide bonds: Link amino acids in proteins (primary structure).
- Glycosidic bonds: Link monosaccharides in carbohydrates.
- Phosphodiester bonds: Link nucleotides in DNA and RNA.
- Disulfide bonds: Covalent bonds between the sulfur atoms of two cysteine residues; stabilize tertiary and quaternary protein structure.
- Ester and Thioester bonds: Found in lipids and metabolic intermediates.
Ionic Bonds
Definition: Formed when one atom donates one or more electrons to another, creating oppositely charged ions (a positive cation and a negative anion) that are held together by strong electrostatic attraction.
Significance: While less common than covalent bonds in the backbones of biomolecules, they are crucial for specific structural and functional roles.
Examples in Biology:
- Found in biological salts.
- Interactions between charged amino acid side chains in proteins (salt bridges).
- Interaction between the negatively charged phosphate groups of DNA and positively charged proteins (e.g., histones).
- Binding of metal ion cofactors to enzymes.
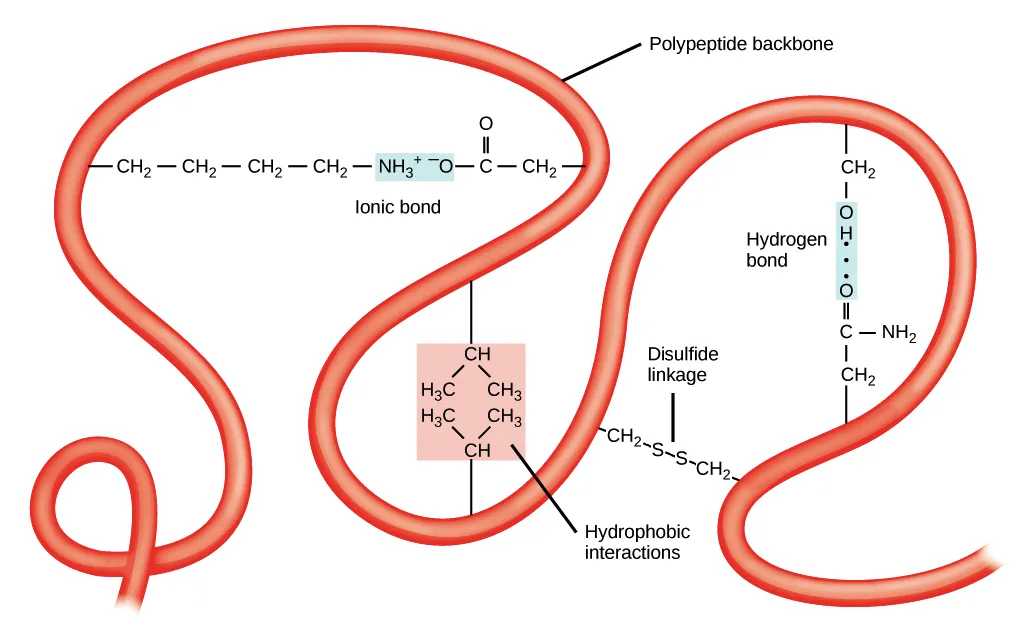
Intermolecular and Intramolecular Interactions
These are weaker forces that are collectively essential for the three-dimensional structure and function of biomolecules.
Hydrogen Bonding
Definition: A weak attraction between a hydrogen atom (covalently bonded to a highly electronegative atom like O, N, or F) and a lone pair of electrons on a neighboring electronegative atom.
Significance: Crucial for the specific pairing of molecules and stabilizing large structures.
Examples:
- DNA Structure: Holds the two strands of the DNA double helix together. There are two H-bonds between Adenine (A) and Thymine (T), and three H-bonds between Guanine (G) and Cytosine (C).
- Protein Structure: Stabilizes secondary structures like alpha-helices and beta-sheets.
- Protein–DNA Interactions: Allows proteins to recognize and bind to specific DNA sequences.
Hydrophobic Interactions
Definition: The tendency of nonpolar (water-fearing) molecules or parts of molecules to aggregate together in an aqueous environment to minimize their contact with polar water molecules.
Significance: A major driving force in the folding of macromolecules and the formation of biological membranes.
Examples:
- Protein Folding: Nonpolar amino acids are buried in the protein's core, away from the surrounding water.
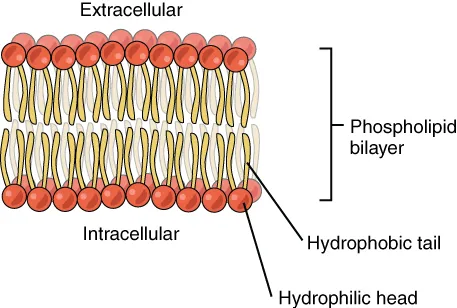
- Membrane Formation: The hydrophobic tails of phospholipids face inward, forming the core of the cell membrane, while the hydrophilic heads face the aqueous environment.
- Ligand Binding: Nonpolar ligands can bind to hydrophobic pockets in proteins.
Hydrophilic Interactions
Definition: The attraction between polar (water-loving) molecules and water. These molecules have partial positive and negative charges that can interact with the polar water molecules.
Significance: Ensures that polar and charged molecules remain dissolved and functional in the aqueous environment of the cell.
Examples:
- Protein Solubility: Hydrophilic amino acid residues on the surface of proteins interact with water, keeping the protein dissolved and stable.
- Cell Membrane Structure: The hydrophilic phosphate heads of phospholipids interact with the water inside and outside the cell.
- Nucleic Acid Solubility: The negatively charged phosphate backbone of DNA and RNA is hydrophilic, allowing these molecules to function in the aqueous nucleoplasm and cytoplasm.
- Enzyme Active Sites: Hydrophilic active sites can interact with polar substrates and water to catalyze biochemical reactions.
Van der Waals Forces
Definition: Weak, short-range attractions between nonpolar molecules due to temporary dipoles caused by the movement of electrons.
Significance: Although individually weak, they contribute significantly to the stability of the interior of proteins and the stacking of base pairs in DNA when many such interactions occur simultaneously.
Summary Table
| Interaction Type | Description | Relative Strength | Biological Example |
|---|---|---|---|
| Covalent Bond | Sharing of electron pairs | Strongest | Peptide bonds in proteins |
| Ionic Bond | Attraction of opposite charges | Strong | Salt bridges in proteins |
| Hydrogen Bond | H atom shared between electronegative atoms | Weak | DNA base pairing (A–T, G–C) |
| Hydrophobic Interaction | Clustering of nonpolar groups away from water | Weak | Core of a folded protein |
| Hydrophilic Interaction | Attraction of polar groups to water | Weak | Phospholipid heads in membrane |
| Van der Waals Forces | Temporary dipole attractions | Weakest | Base stacking in DNA |
<PracticeQuestions questionsString="[{"question":"What is the primar