2.19 Molecular Structure of Acylglycerol, Phospholipid and Terpene
This section details the structure, properties, and classification of major lipid groups, including acylglycerols (fats and oils), waxes, phospholipids, and terpenes. Lipids are a diverse group of organic molecules that are largely nonpolar and hydrophobic.
Key Concepts
Acylglycerols (Triglycerides)
Acylglycerols are the most abundant lipids in living organisms and serve primarily as energy storage molecules.
Definition: They are esters formed from a molecule of glycerol and one, two, or three fatty acid molecules.
Formation (Esterification): The chemical reaction between an alcohol (glycerol's -OH group) and a carboxylic acid (fatty acid's -COOH group), which forms an ester linkage (-COO-) and releases a water molecule.
- Glycerol: A simple 3-carbon alcohol with a hydroxyl (-OH) group on each carbon.
- Fatty Acid: A long hydrocarbon chain (R) with a carboxyl group (-COOH) at one end.
[FC: fc_3r8lyj]
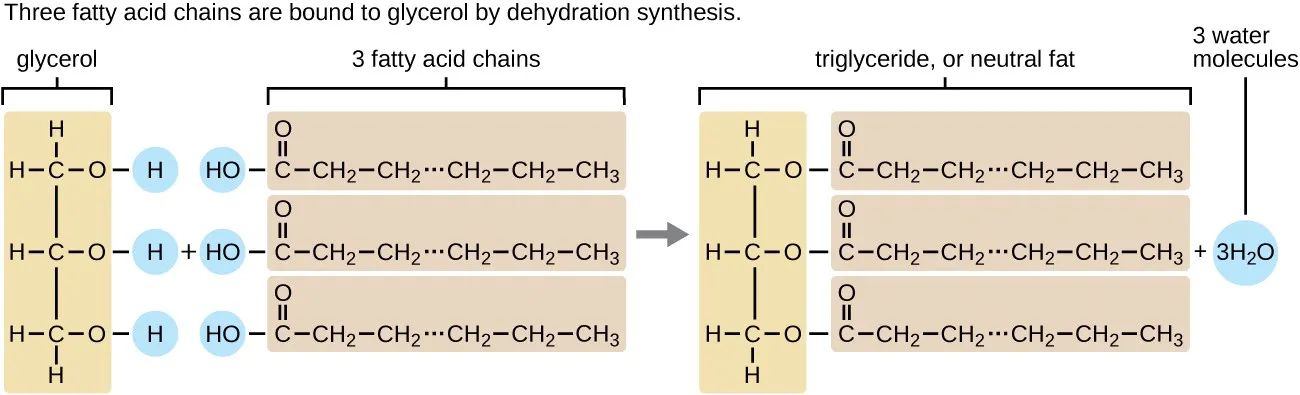 Figure 2.30: Formation of a triacylglycerol (neutral lipid)
Figure 2.30: Formation of a triacylglycerol (neutral lipid)
Classification:
- Monoacylglycerol: 1 glycerol + 1 fatty acid.
- Diacylglycerol: 1 glycerol + 2 fatty acids.
- Triacylglycerol (Triglyceride): 1 glycerol + 3 fatty acids. These are also called neutral lipids because all polar -OH groups of glycerol are esterified, leaving no net charge.
[FC: fc_8p922z]
Properties and Types of Fatty Acids
Fatty acids are the building blocks of many lipids and vary in the length of their hydrocarbon chains and the presence of double bonds.
| Property Comparison | Saturated Fatty Acids | Unsaturated Fatty Acids |
|---|---|---|
| Carbon Bonds | Only single C-C bonds in the hydrocarbon tail. | One or more C=C double bonds in the tail. |
| Hydrogenation | "Saturated" with the maximum possible number of hydrogen atoms. | Not fully saturated with hydrogen atoms due to double bonds. |
| State at Room Temp. | Solid (e.g., animal fats like butter). | Liquid (e.g., plant oils like olive oil). |
| Melting Point | Higher | Lower |
| Common Source | Animal lipids | Plant lipids |
| Example | Palmitic acid, Stearic acid | Oleic acid, Linoleic acid |
Table 2.6: Common types of fatty acids
| Name | Typical Source | No. of Carbons | Condensed Formula | Melting point (°C) |
|---|---|---|---|---|
| Saturated | ||||
| 1. Lauric acid | Coconut oil | 12 | 44 | |
| 2. Myristic acid | Butter fat | 14 | 58 | |
| 3. Palmitic acid | Most fats and oils | 16 | 63 | |
| 4. Stearic acid | Most fats and oils | 18 | 70 | |
| Unsaturated | ||||
| 5. Oleic acid | Olive oil | 18 | 4 | |
| 6. Linoleic acid | Vegetable oils | 18 | -5 | |
| 7. Linolenic acid | Soybeans and canola oils | 18 | -11 | |
| 8. Arachidonic acid | Poultry, fish, egg | 20 | -50 |
[FC: fc_x86za6]
[MCQ: 5819967b-7ee7-4fde-a1dc-080fb0fe7045]
[MCQ: 6385fed5-507e-452f-8cdc-1df09330c6f3]
Waxes
Definition: Highly hydrophobic, chemically inert lipids with protective functions.
Types:
- Natural Waxes: Esters of long-chain fatty acids and long-chain alcohols.
- Examples: Bee's wax (in honeycombs), cutin (on plant leaf surfaces to prevent water loss).
- Synthetic Waxes: Derived from petroleum.
- Example: Paraffin wax (used in candles).
Phospholipids
Phospholipids are crucial structural components of cell membranes.
Structure: Derived from phosphatidic acid, which consists of a glycerol molecule, two fatty acids, and a phosphate group.
Formation: A phospholipid is formed when a phosphatidic acid molecule combines with a small organic group (e.g., choline, serine, ethanolamine).
Amphipathic Nature: Phospholipids have both hydrophilic and hydrophobic regions.
- Hydrophilic Head: The phosphate group and its attachment (e.g., choline) are polar and water-soluble.
- Hydrophobic Tails: The two fatty acid chains are nonpolar and water-insoluble.
Example: Phosphatidylcholine (also known as Lecithin) is a common phospholipid in cell membranes.
[FC: fc_ijz1qo]
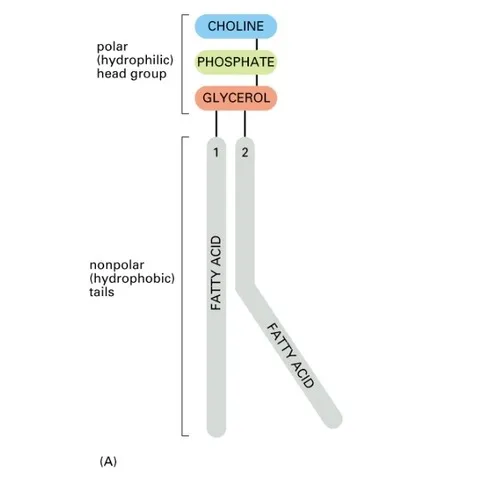 Figure 2.31: Phosphatidylcholine (Lecithin)
Figure 2.31: Phosphatidylcholine (Lecithin)
Terpenes
Structure: Synthesized from a five-carbon building block called the isoprene unit.
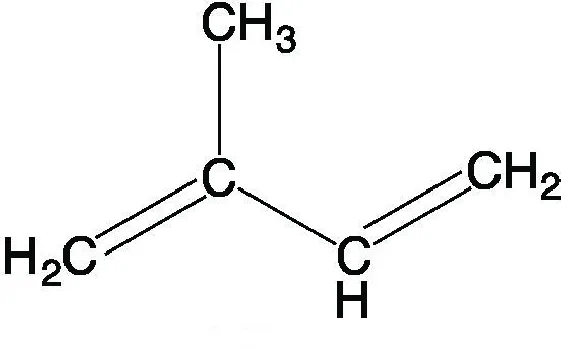 Figure 2.32: Isoprene unit
Figure 2.32: Isoprene unit
Classification and Examples: Terpenes are formed by the condensation of multiple isoprene units.
- Monoterpene (2 units): Menthol
- Diterpene (4 units): Vitamin A, phytol (part of chlorophyll)
- Triterpene (6 units): Ambrein
- Polyterpene (>6 units): Natural rubber
[FC: fc_k32ki3]
[MCQ: 07059480-8f54-4035-95a7-c65edd4db386]
Possible Questions and Answers
Q: What is the difference between a fat and an oil?
A: Both are triacylglycerols. Fats are solid at room temperature and are rich in saturated fatty acids (common in animals). Oils are liquid at room temperature and are rich in unsaturated fatty acids (common in plants).
Q: Why are phospholipids essential for cell membranes?
A: Their amphipathic nature (having a hydrophilic head and hydrophobic tails) allows them to spontaneously form a lipid bilayer in an aqueous environment. This bilayer is the fundamental structure of all cell membranes, creating a barrier between the cell's interior and exterior.
Q: What defines a fatty acid as "saturated" or "unsaturated"?
A: A saturated fatty acid has no carbon-carbon double bonds in its hydrocarbon tail, meaning it holds the maximum number of hydrogen atoms possible. An unsaturated fatty acid has one or more carbon-carbon double bonds, creating "kinks" in the chain and reducing the number of hydrogen atoms.
Summary
- Lipids are a diverse group of hydrophobic organic molecules. Key types include acylglycerols, waxes, phospholipids, and terpenes.
- Acylglycerols (fats and oils) are esters of glycerol and fatty acids, primarily used for energy storage. They are formed via esterification.
- Fatty Acids can be saturated (no C=C bonds, solid at room temp) or unsaturated (one or more C=C bonds, liquid at room temp).
- Waxes are simple lipids with protective, water-repellent functions in both plants (cutin) and animals (bee's wax).
- Phospholipids are amphipathic molecules (hydrophilic head, hydrophobic tails) that form the structural basis of cell membranes.
- Terpenes are lipids built from repeating five-carbon isoprene units and include compounds like Vitamin A, chlorophyll components, and natural rubber.
The diverse structures of lipids lead to a wide array of functions, from long-term energy storage and thermal insulation (acylglycerols) to forming essential biological barriers (phospholipids) and serving as vitamins and pigments (terpenes).
References
(Derived from FBISE textbook)