8.5 Carnot Heat Engine and Carnot Cycle
The Carnot Heat Engine is a theoretical, idealized thermodynamic engine proposed by Sadi Carnot in 1824. It is not a practical engine but a conceptual model that operates on a reversible cycle known as the Carnot Cycle. Its importance lies in setting the maximum possible efficiency that any heat engine can achieve when operating between two given temperatures.
Construction of the Ideal Engine
The theoretical engine consists of:
- A cylinder with a frictionless, movable piston.
- The walls of the cylinder and the piston are perfect insulators, preventing heat transfer.
- The base of the cylinder is a perfect conductor, allowing heat to be exchanged with external reservoirs.
- The working substance is typically an ideal gas.
The Carnot Cycle
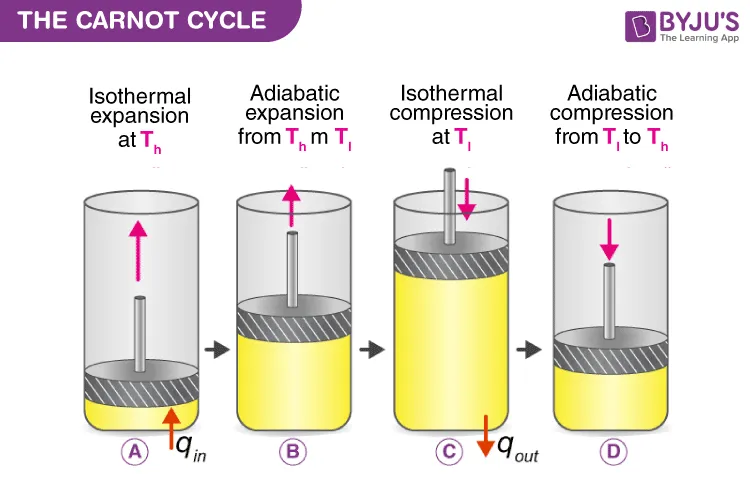
The Carnot Cycle is a sequence of four reversible processes that return the working substance to its original state. The cycle involves interaction with a high-temperature hot reservoir () and a low-temperature cold reservoir ().
The four steps are:
-
Isothermal Expansion (A → B): The cylinder is placed on the hot reservoir at temperature . The gas expands slowly, doing work on the piston while absorbing heat () from the reservoir to keep its temperature constant.
-
Adiabatic Expansion (B → C): The cylinder is placed on an insulating stand. The gas continues to expand and do work, but with no heat exchange. As a result, its internal energy decreases, and its temperature drops from to .
-
Isothermal Compression (C → D): The cylinder is placed on the cold reservoir at temperature . An external force compresses the gas, and as work is done on it, the gas rejects heat () to the cold reservoir to keep its temperature constant.
-
Adiabatic Compression (D → A): The cylinder is returned to the insulating stand. The gas is further compressed with no heat exchange. Work is done on the gas, which increases its internal energy and raises its temperature from back to the initial temperature .
The PV Diagram for the Carnot Cycle
On a Pressure-Volume (PV) diagram, the Carnot cycle forms a closed loop. The area enclosed by this loop represents the net work done () by the engine in one cycle.
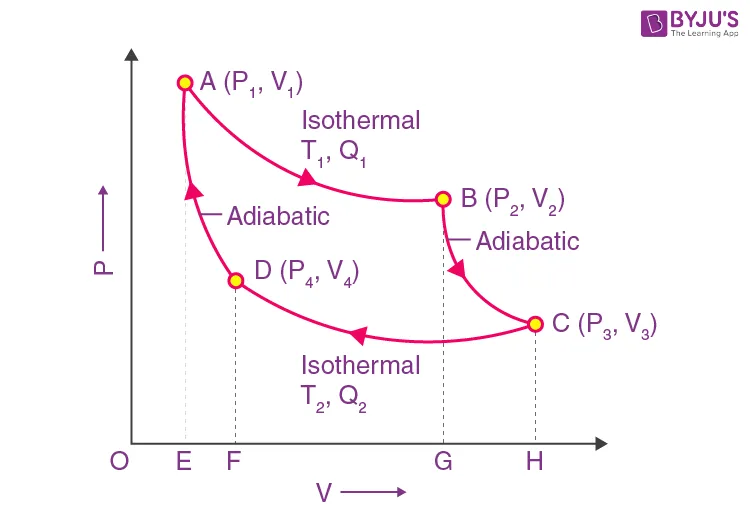
| Stage | Process | Heat Transfer | Temperature |
|---|---|---|---|
| A → B | Isothermal Expansion | Absorbs heat | Constant at |
| B → C | Adiabatic Expansion | None () | Drops from to |
| C → D | Isothermal Compression | Rejects heat | Constant at |
| D → A | Adiabatic Compression | None () | Rises from to |
Efficiency of the Carnot Engine
The thermal efficiency () of any heat engine is the ratio of the net work done () to the heat absorbed from the hot reservoir ().
From the First Law of Thermodynamics for a cycle, the net work done is the difference between the heat absorbed and the heat rejected: .
For the ideal, reversible Carnot cycle, it can be shown that the ratio of heat exchanged is equal to the ratio of the absolute temperatures of the reservoirs.
This gives the famous formula for the efficiency of a Carnot engine:
Carnot's Theorem
This theorem establishes the significance of the Carnot engine. It states:
- No heat engine operating between two given temperature reservoirs can be more efficient than a reversible Carnot engine operating between the same two reservoirs.
- All reversible heat engines operating between the same two temperature reservoirs have the same efficiency.
Possible Questions and Answers
Q: Is it possible for a Carnot engine to have 100% efficiency?
A: No. According to the formula , 100% efficiency () would only be possible if the cold reservoir temperature () was at absolute zero (0 Kelvin), which is physically unattainable.
Q: Why is the Carnot engine only a theoretical model?
A: The Carnot cycle requires processes that are perfectly reversible and infinitely slow to maintain thermal equilibrium (isothermal steps) and perfectly insulated (adiabatic steps). These ideal conditions, such as frictionless pistons, perfect insulators, and infinitely slow processes, cannot be achieved in the real world.