7.1 Classification of Solids
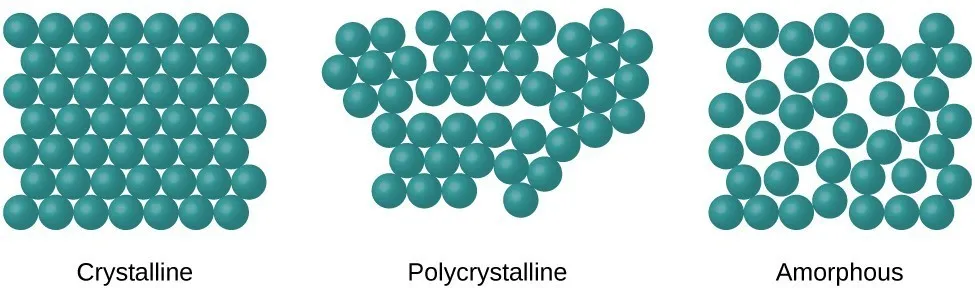
Solids are a state of matter characterized by a fixed shape and volume, making them incompressible. The distinct properties of different solids arise from the arrangement of their constituent atoms, ions, or molecules. Based on this internal atomic structure, solids are broadly classified into three main types: crystalline, polycrystalline, and amorphous.
For more on how solids respond to external forces, see Deformation In Solids→.
Crystalline Solids
Crystalline solids are defined by a highly ordered, three-dimensional arrangement of atoms, ions, or molecules. This regular, repeating pattern, known as a crystal lattice, extends throughout the entire material.
- Properties:
- Have a definite geometric shape with flat faces and sharp edges.
- Possess a sharp, well-defined melting point.
- Exhibit anisotropic behavior, meaning their physical properties (like electrical conductivity or refractive index) vary with the direction of measurement.
- Produce clean, flat surfaces when cut (cleavage).
- Examples: Salts (), metals (copper, iron), diamond, sulfur, quartz.
Polycrystalline Solids
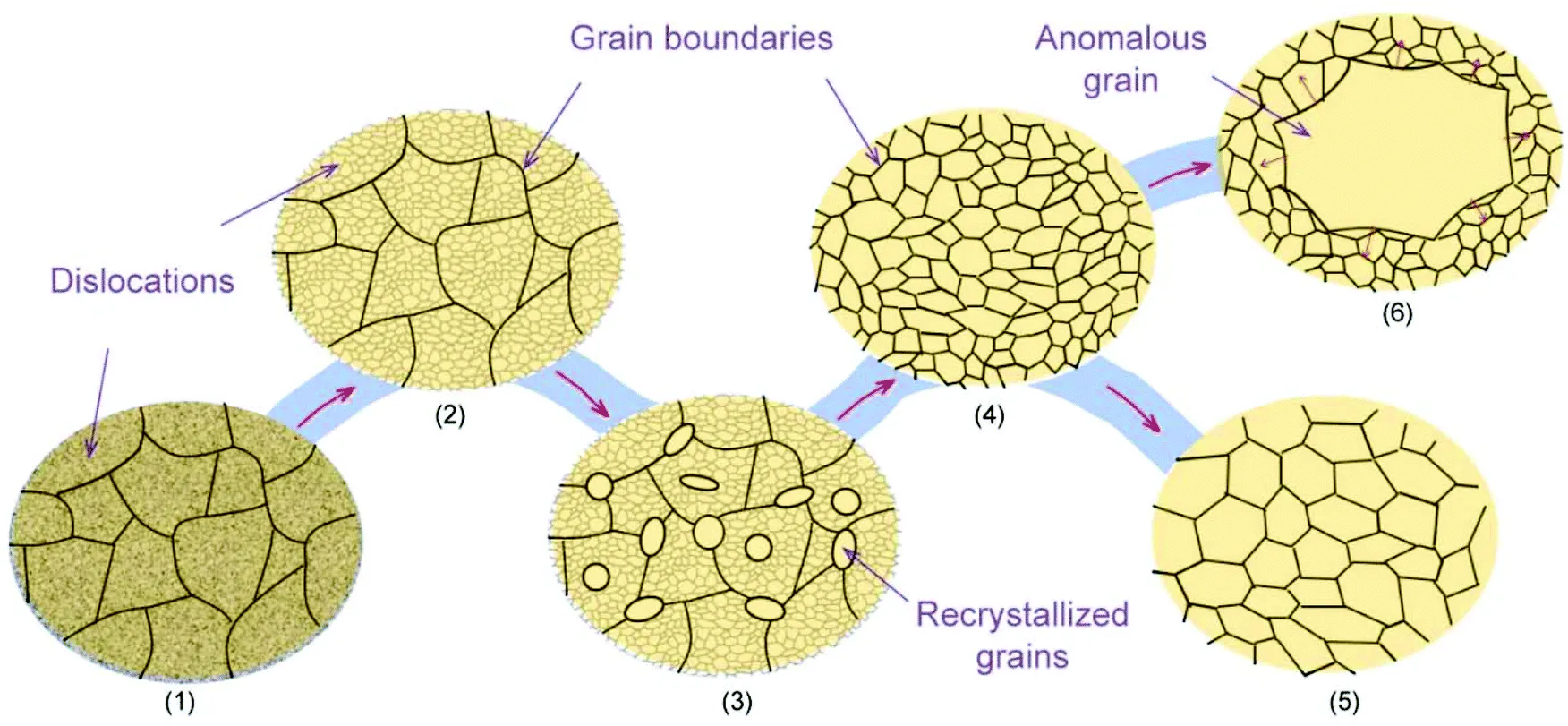
Polycrystalline solids are composed of many small, individual crystals, known as grains or crystallites. While each grain has an ordered crystalline structure, the grains themselves are randomly oriented with respect to each other.
-
Properties:
- Appear uniform on a macroscopic scale.
- Exhibit isotropic properties, meaning their physical properties are the same in all directions, due to the random orientation of the grains.
- Generally possess good strength and toughness.
-
Examples: Most metals in their standard form, ceramics, and many polymers like rubber and polythene.
- Polymers are large molecules formed by the linking of repeating smaller units (monomers) through a process called polymerization.
Amorphous Solids
Amorphous (or "non-crystalline") solids lack any long-range ordered atomic structure. Their atoms are arranged randomly, much like in a liquid that has been "frozen" in place. They are often called glassy solids.
-
Properties:
- Do not have a definite geometric shape.
- Do not have a sharp melting point; they soften gradually over a range of temperatures.
- Are isotropic, exhibiting uniform properties in all directions.
- Produce irregular, often curved surfaces when broken (fracture).
-
Examples: Glass, rubber, glues, and many plastics.
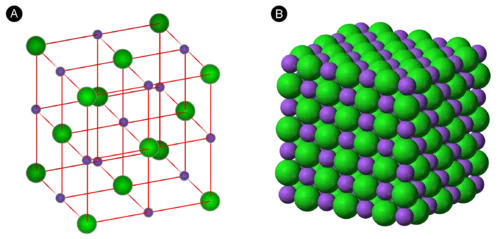
Unit Cell and Crystal Lattice
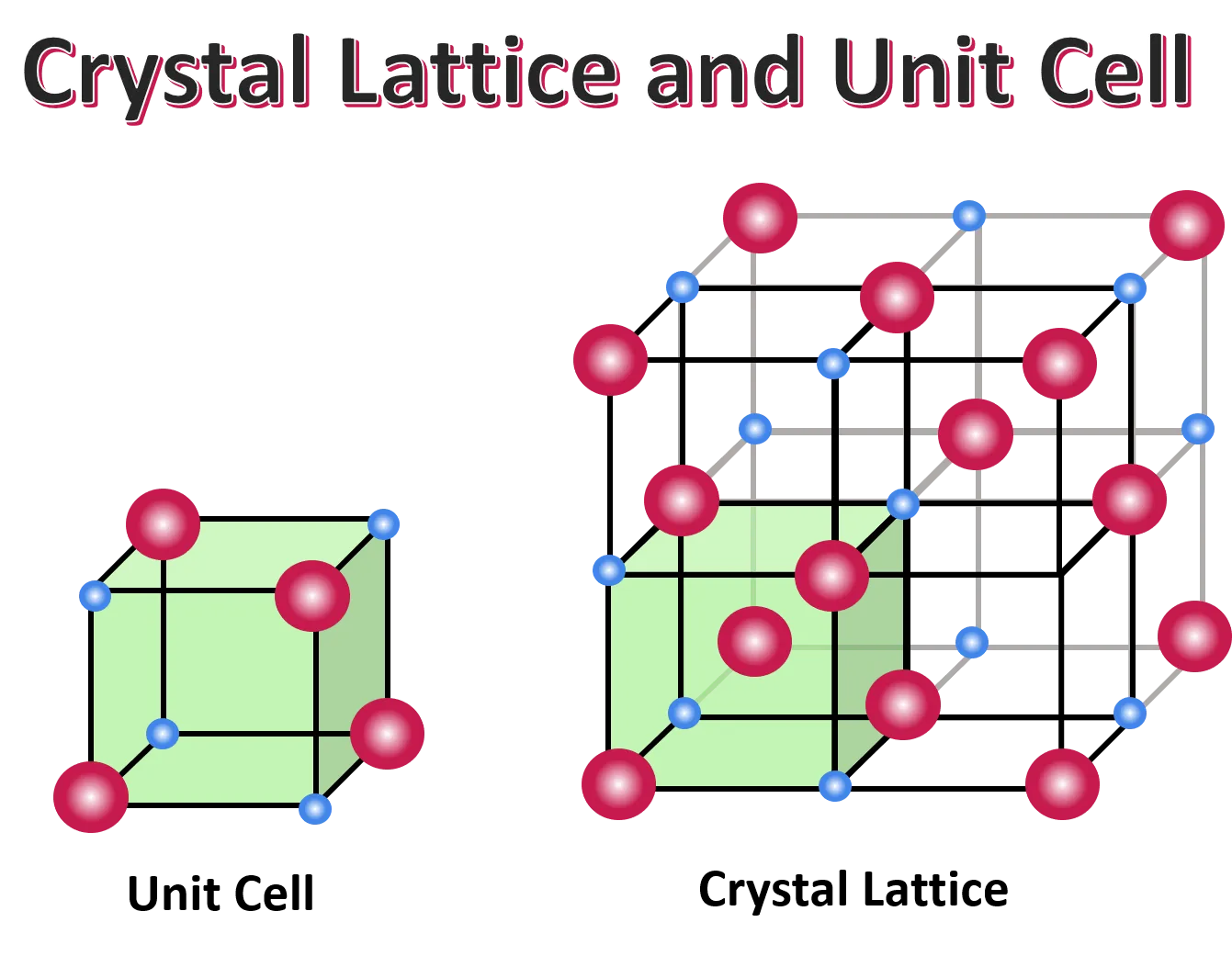
The ordered structure of a crystalline solid is described by its crystal lattice, an imaginary framework of points representing the positions of atoms, ions, or molecules.
- Unit Cell: The smallest repeating unit of a crystal lattice that, when stacked in three dimensions, forms the entire crystal.
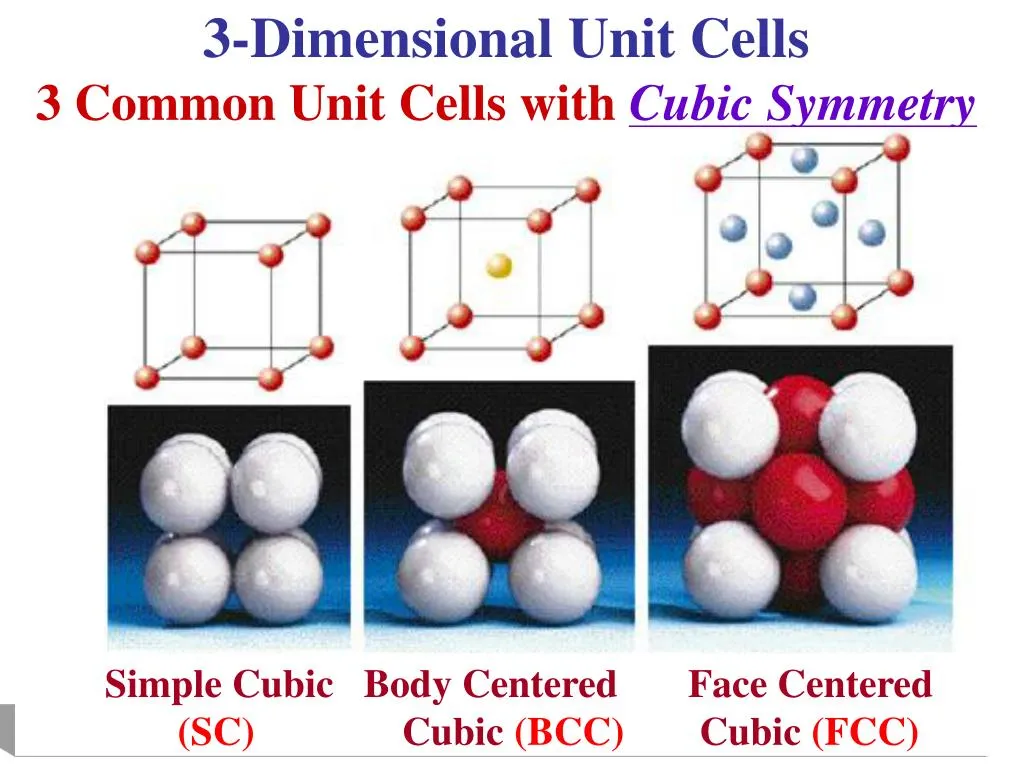
- Types of Cubic Unit Cells:
- Simple Cubic (SC): One atom per unit cell.
- Body-Centered Cubic (BCC): Two atoms per unit cell.
- Face-Centered Cubic (FCC): Four atoms per unit cell.
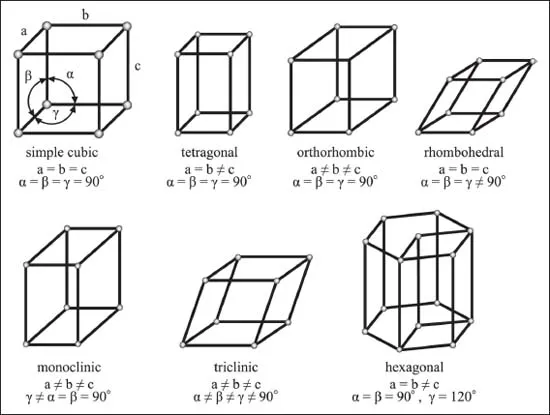
- Unit Cell Dimensions: A unit cell is defined by the lengths of its three sides (, , ) and the angles between them (, , ). There are seven fundamental shapes for unit cells.
Possible Questions and Answers
Q: What is the difference between anisotropic and isotropic?
A: Anisotropic means that a material's properties depend on the direction in which they are measured (characteristic of single crystals). Isotropic means the properties are the same in all directions (characteristic of amorphous and polycrystalline solids).
Q: Why is glass considered an amorphous solid and not a true solid?
A: Glass lacks the long-range ordered atomic structure of a crystalline solid. Its random atomic arrangement is like that of a liquid, which is why it is sometimes called a "super-cooled liquid." It softens over a temperature range rather than melting at a specific point.
| Property | Crystalline | Polycrystalline | Amorphous |
|---|---|---|---|
| Atomic Structure | Ordered, long-range | Ordered in small grains | Disordered, random |
| Melting Point | Sharp, definite | Definite | Softens over a range |
| Directional Properties | Anisotropic | Isotropic | Isotropic |
| Examples | Salt, Diamond | Metals, Ceramics | Glass, Rubber |