22.5 Acid Rain
Introduction to Acid Rain
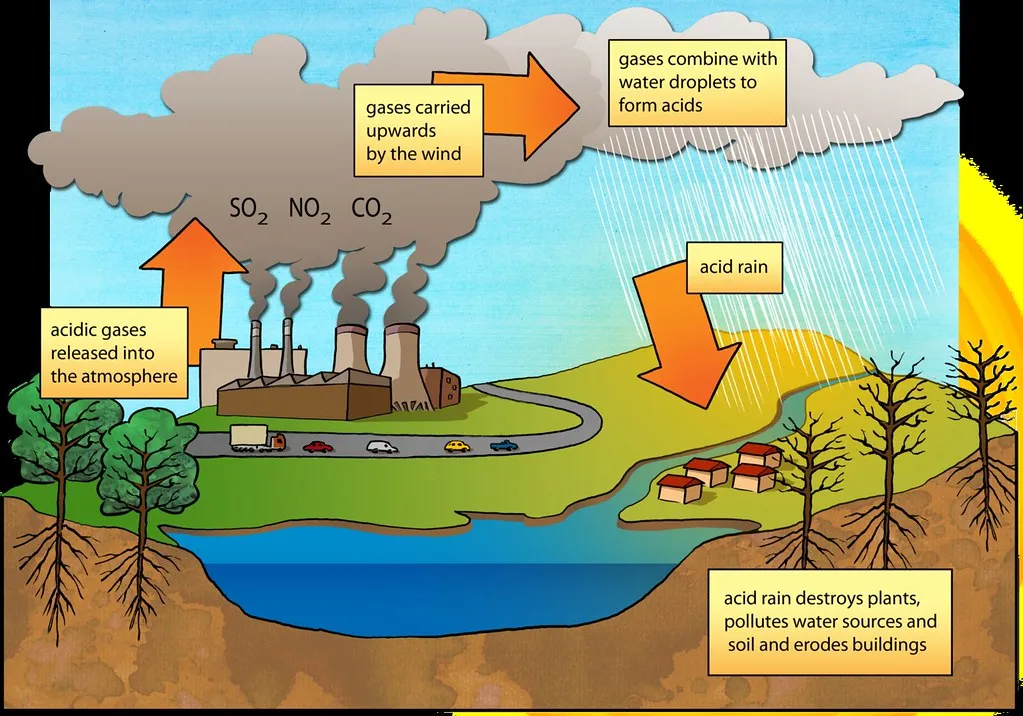
Normal rainwater, being saturated with carbon dioxide, typically has a pH of 5.6. Rain is classified as acid rain when its pH falls below this value. Acid rain is a direct consequence of air pollution.
Causes of Acid Rain
The primary atmospheric pollutants responsible for acid rain are:
- Sulphur oxides (): Primarily sulfur dioxide () and sulfur trioxide (), often released from the burning of fossil fuels such as coal in power plants and industrial processes.
- Nitrogen oxides (): Such as nitric oxide () and nitrogen dioxide (), largely produced from high-temperature combustion processes in vehicles and industrial facilities.
These oxides dissolve in rainwater and react to form strong acids:
-
Formation of Sulfuric Acid:
-
Formation of Nitric Acid:
As a result of these reactions, rainwater pH can drop significantly, sometimes as low as 2.1, which is more acidic than lemon juice or vinegar. When this acid rain falls on plants and soil, it initiates a series of chemical reactions that profoundly affect soil chemistry, nutrient availability, and ultimately, crop growth and yield.
Impact of Acid Rain on Soil and Plants
The effects of acid rain on crops and soil are severe and multifaceted.
1. Lowering Soil pH and Nutrient Leaching
Acid rain significantly lowers the pH of the soil, making it more acidic. This increased acidity has detrimental effects on soil composition:
-
Nutrient Displacement: Important plant nutrients such as calcium (), magnesium (), potassium (), and sodium () are positively charged ions that are typically bound to negatively charged soil particles. The hydrogen ions () from acid rain replace these essential nutrients on the soil particles, causing them to leach out of the soil. This reduces the availability of these vital nutrients to plants.
Example: Reaction of sulfuric acid with calcium carbonate, a common soil component that acts as a buffer:
This reaction consumes the buffering capacity of the soil and releases calcium into a soluble form that can be leached away. This process is also responsible for the erosion of marble monuments, often called "stone leprosy."
2. Aluminium Leaching and Toxicity
Increased soil acidity also causes the leaching of aluminium from naturally occurring aluminium compounds in the soil.
-
Aluminium Dissolution: Aluminium oxide, a common constituent of soil, can react with sulfuric acid:
-
Toxicity to Plants and Aquatic Life: Aluminium ions are highly poisonous. In soil, they damage plant roots and inhibit nutrient uptake. When leached into lakes, they cause mucus buildup on fish gills, leading to suffocation.
3. Direct Damage to Plant Foliage
Acid rain can directly damage the waxy cuticles on the surface of plant leaves. This damage has several negative consequences:
- Increased Water Loss: A compromised waxy layer leads to increased transpiration and water loss from the plant.
- Increased Susceptibility: Plants become more vulnerable to diseases, insect infestations, and environmental stressors such as drought and frost.
4. Impact on Sensitive Crops
Studies have shown that acid rain can significantly affect the performance of sensitive agricultural crops:
- Physiological and Biochemical Changes: Crops such as soybeans, wheat, and peanuts are particularly sensitive to changes in soil pH and direct acid exposure.
- Reduced Nitrogen Fixation: In legumes like soybeans, acid rain can reduce nitrogen fixation, a crucial process for their growth.
5. Effects on Soil Microbial Communities
Soil microbial communities, including bacteria and fungi, play a critical role in maintaining soil health through nutrient cycling and decomposition. Acid rain negatively affects these microbial communities by altering their optimal pH range, further impacting nutrient availability and soil structure.