19.1 Main Components of Chromatography
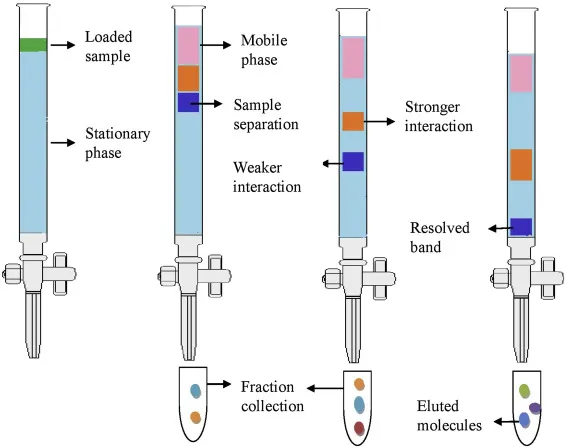
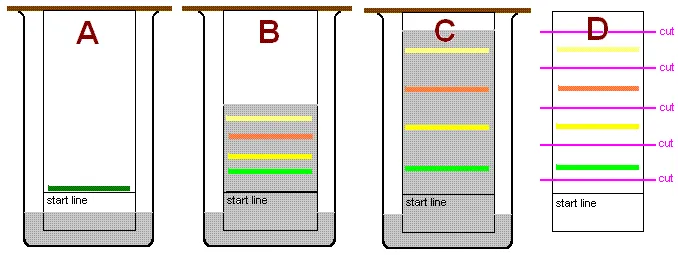
Chromatography is a laboratory technique for the separation of a mixture. The mixture is dissolved in a fluid called the mobile phase, which carries it through a structure holding another material called the stationary phase. The various constituents of the mixture travel at different speeds, causing them to separate.
1. Stationary Phase
The stationary phase is the component of the chromatographic system that remains fixed in place. The mobile phase moves over it, and the separation occurs based on the differential interactions of the mixture's components with this phase.
- Nature: It can be a solid (e.g., silica, glass) or a liquid coated onto the surface of a solid support.
- Function: In column chromatography, the stationary phase is typically packed into a column. As the mobile phase flows through, components that interact more strongly with the stationary phase move more slowly, leading to separation.
Support
In many chromatographic techniques, the stationary phase is held by a support. This is an inert material that provides a surface for the stationary phase to adhere to without reacting with the solutes.
2. Mobile Phase
The mobile phase is the solvent (liquid or gas) that flows through the stationary phase, carrying the components of the mixture with it.
- Liquid Chromatography (LC): Uses a liquid mobile phase.
- Gas Chromatography (GC): Uses a gas mobile phase.
- Function: The mobile phase dissolves the sample and carries it through the column. The separation depends on how the components partition between the mobile and stationary phases. There is a dynamic equilibrium as components constantly move between the two phases.
For more on selecting appropriate mobile and stationary phases, see Selection of Mobile and Stationary Phase→.
3. Retardation Factor () Value
The Retardation Factor () is a dimensionless quantity used in planar chromatography (like paper or thin-layer chromatography) to identify compounds. It represents the ratio of the distance traveled by a solute (compound) to the distance traveled by the solvent front.
- The value is characteristic of a specific compound under a given set of conditions (stationary phase, mobile phase, temperature).
- It is calculated using the following formula:
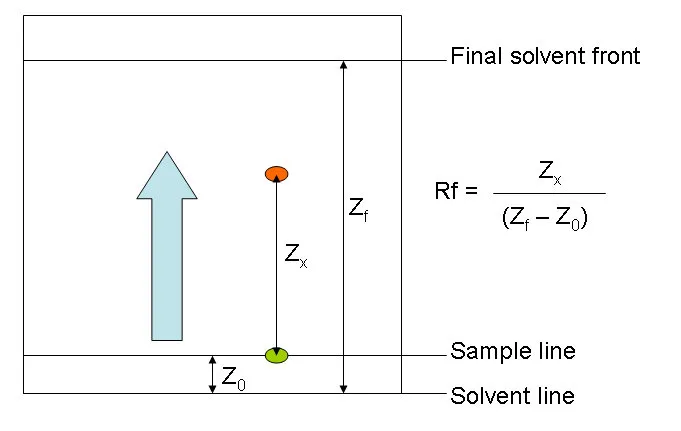
For detailed interpretation of values, refer to .
4. Solvent Front
The solvent front is the furthest point reached by the mobile phase (solvent) as it moves up the stationary phase (e.g., chromatography paper). This distance is used as the denominator in the calculation.
5. The Baseline
The baseline is the starting line where the initial sample mixture is applied (spotted) on the stationary phase. All distance measurements for calculating the value begin from this reference point.
Applications of Chromatography
- Biochemical Research: Used for the separation and identification of complex biological compounds like proteins, amino acids, and nucleic acids.
- Petroleum Industry: Employed to analyze complex mixtures of hydrocarbons in crude oil and refined products.
- Pharmaceuticals: Essential for quality control, drug discovery, and ensuring the purity of medicines.
- Environmental Science: Used to detect and quantify pollutants in air, water, and soil samples.