18.6 Atomic Absorption Spectrum
An atomic absorption spectrum is produced when free, gaseous atoms in their ground state absorb light of specific wavelengths. This absorption of energy causes the atoms' electrons to get excited and jump to higher energy levels. The spectrum shows dark lines at the exact wavelengths where the light was absorbed.
Process
- A light source containing a wide range of wavelengths is passed through a sample of free atoms (e.g., in a flame or furnace).
- Atoms in the sample absorb photons of very specific energies (wavelengths) that correspond to the energy differences between their electronic states.
- This absorption excites the electrons from the ground state to a higher energy state.
- A detector on the other side of the sample records the transmitted light, revealing a spectrum with dark lines where absorption occurred.
Characteristic: Atomic absorption spectra are always line spectra, meaning they consist of discrete lines rather than a continuous band of colors.
Comparison with Atomic Emission Spectrum (AES)
Atomic emission and absorption spectra for a given element provide the same information about its electronic structure, as the wavelengths of light absorbed (in AAS) are the same as those emitted (in AES).
For more details on atomic emission, see 18.5 Atomic Emission Spectroscopy→(/chemistry-12/18-spectroscopy/18-5-atomic-emission-spectroscopy).
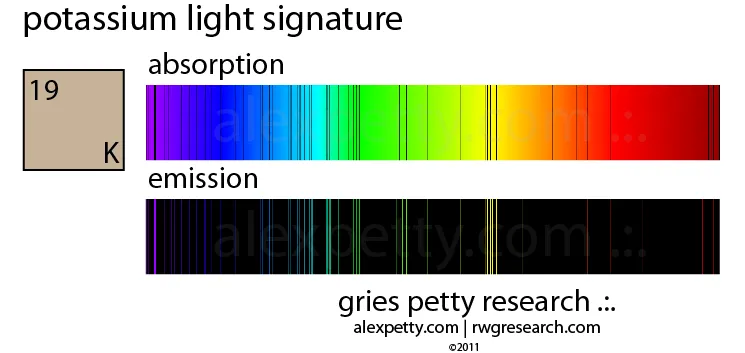
The primary difference between the two is their appearance, as summarized in the table below:
| Feature | Atomic Absorption Spectrum (AAS) | Atomic Emission Spectrum (AES) |
|---|---|---|
| Process | Atoms absorb energy (photons). | Excited atoms release energy (photons). |
| Appearance | Dark lines on a bright, continuous background. | Bright lines on a dark background. |
| Information | Reveals the wavelengths of light an element can absorb. | Reveals the wavelengths of light an element can emit. |
| Relationship | It is the complementary spectrum to the emission spectrum. | It is the complementary spectrum to the absorption spectrum. |
Possible Questions/Answers
Q: What causes the dark lines in an atomic absorption spectrum?
A: The dark lines are caused by atoms in their ground state absorbing light of specific wavelengths to become excited. These wavelengths are thus missing from the light that passes through the sample.
Q: Why do the lines in the absorption and emission spectra for potassium appear at the same wavelengths?
A: Because the energy transitions of electrons are quantized and specific for each element. The energy absorbed for an electron to jump from a lower to a higher level is exactly the same as the energy released when it falls back down between those same two levels.
Significance
Atomic absorption spectroscopy is a powerful analytical technique used to determine the concentration of a specific element in a sample, with applications in environmental testing, clinical analysis, and industry. It follows the Beer-Lambert Law, where absorbance is directly proportional to the concentration of the analyte.