17.6 Deduction of Chlorine and Bromine in a Molecule by Mass Spectrometry
Mass spectrometry is a powerful analytical technique that can identify the presence of certain elements in a molecule based on their isotopic abundances. Chlorine and bromine, in particular, have distinctive isotopic patterns that produce characteristic peaks in a mass spectrum, making them easily identifiable.
The Role of Isotopes in Mass Spectrometry
Many elements exist naturally as a mixture of isotopes, atoms with the same number of protons but different numbers of neutrons, resulting in different masses. Mass spectrometry separates ions based on their mass-to-charge ratio (). The presence of heavier isotopes leads to additional peaks in the mass spectrum, typically at higher values than the main molecular ion peak (). A peak appearing two mass units higher than the molecular ion peak is called the M+2 peak.
1. Deduction of Chlorine (Cl)
Chlorine has two stable isotopes, and , with significant natural abundances.
-
Isotopes and Abundances:
- : Relative abundance of 75.77%
- : Relative abundance of 24.23%
-
Mass Spectrum Pattern:
- A molecule containing one chlorine atom will show two peaks in the molecular ion region.
- The molecular ion peak () corresponds to the molecule containing the isotope.
- An M+2 peak appears for the molecule containing the heavier isotope.
- The ratio of the heights of the peak to the peak is approximately 3:1, reflecting the natural abundance of the isotopes.
Example: Mass Spectrum of Chloromethane ()
The mass spectrum of chloromethane clearly illustrates this pattern.
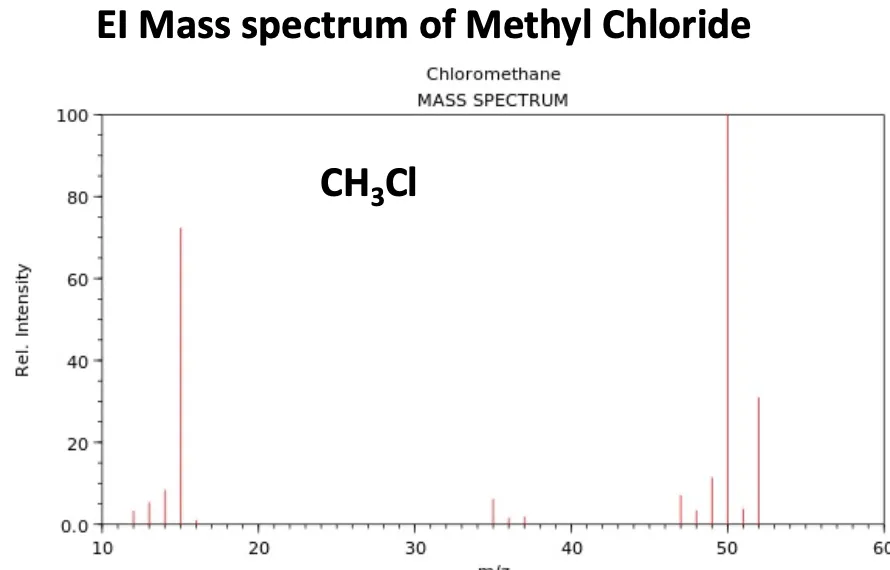
- The molecular ion peak () for appears at .
- The M+2 peak for appears at .
- The height of the peak at is approximately one-third the height of the peak at , confirming the presence of a single chlorine atom.
2. Deduction of Bromine (Br)
Bromine also has two stable isotopes, and , with nearly equal abundances.
-
Isotopes and Abundances:
- : Relative abundance of 50.50%
- : Relative abundance of 49.50%
-
Mass Spectrum Pattern:
- A molecule containing one bromine atom will show two prominent peaks in the molecular ion region.
- The molecular ion peak () corresponds to the molecule with the isotope.
- The M+2 peak corresponds to the molecule with the isotope.
- Because their abundances are almost equal, the and peaks have approximately the same height (a 1:1 ratio).
Example: Mass Spectrum of Bromoethane ()
The mass spectrum of bromoethane provides a classic example of bromine's signature.
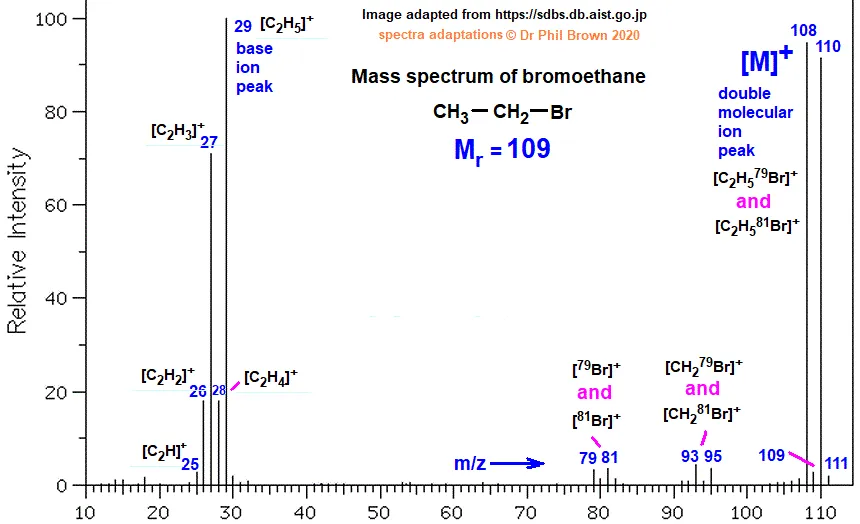
- The molecular ion peak () for appears at .
- The M+2 peak for appears at .
- The heights of these two peaks are nearly identical, which is a definitive indicator of a single bromine atom in the molecule.
Possible Questions and Answers
Q: How can you distinguish between a molecule containing chlorine and one containing bromine using only a mass spectrum?
A: Look at the ratio of the and peaks.
- Chlorine: The peak is about one-third the height of the peak (3:1 ratio).
- Bromine: The peak is approximately the same height as the peak (1:1 ratio).
Q: What causes the M+2 peak in the mass spectrum of a halogenated compound?
A: The M+2 peak is caused by the presence of a heavier, naturally occurring isotope of the halogen (e.g., or ) in the molecule.
| Element | Isotopes | Abundance Ratio (Lighter:Heavier) | Peak Height Ratio ( : M+2) |
|---|---|---|---|
| Chlorine | : | ||
| Bromine | : |
Significance: This technique is crucial in structural elucidation in organic chemistry, helping chemists to quickly determine the presence and number of halogen atoms in an unknown compound.