17.2 Calculation of Relative Atomic Masses
The relative atomic mass () of an element is the weighted average mass of its naturally occurring isotopes relative to 1/12th the mass of a carbon-12 atom. It is calculated using data from mass spectrometry, which provides the mass-to-charge ratio (m/z) and the relative abundance of each isotope.
The relative atomic mass is a weighted average because isotopes occur in different proportions in nature. Simply averaging the masses would give equal importance to each isotope, which does not reflect natural composition.
Mass spectrometry provides two key pieces of information for each isotope:
- The mass number (or m/z value) of the isotope
- The relative abundance of that isotope (as a percentage)
The general formula for calculating the relative atomic mass is:
This formula accounts for the natural abundance of each isotope, giving a weighted average that represents the average mass of an atom of the element as found in nature.
The unit of relative atomic mass is atomic mass unit (amu), but since it is a ratio, it is a dimensionless quantity.
Worked Examples
Example 1: Boron
The mass spectrum for Boron (B, atomic number Z = 5) shows two isotopes. For more details on analyzing isotopes and their abundances, refer to .
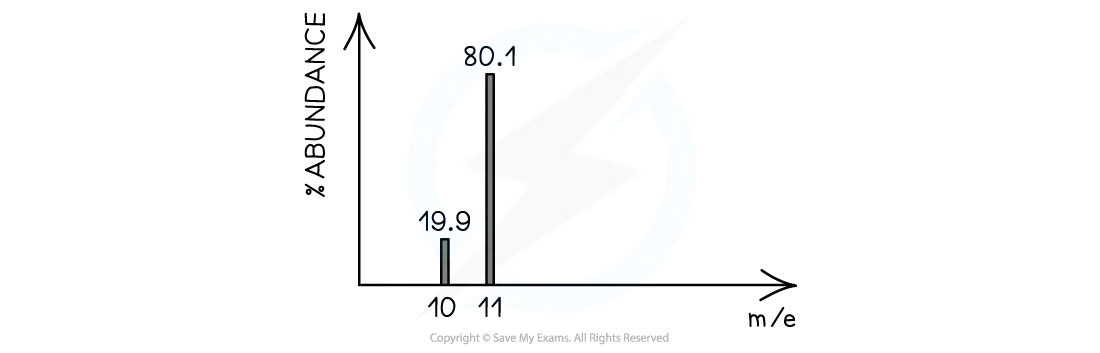
Given Values:
| Isotope | Mass Number | Relative Abundance (%) |
|---|---|---|
| Boron-10 | 10 | 19.90% |
| Boron-11 | 11 | 80.10% |
Calculation:
- Apply the formula:
- Substitute the values:
- Calculate the result:
The calculated relative atomic mass of Boron is 10.801 amu.
Practice Question
Concept Assessment Exercise 17.1
Q: Using the given mass spectrum, calculate the average atomic mass of the element and identify it using the periodic table.

A:
-
Extract data from the spectrum:
- Isotope 1: Mass = 20, Relative Abundance ≈ 90.9%
- Isotope 2: Mass = 21, Relative Abundance ≈ 0.2%
- Isotope 3: Mass = 22, Relative Abundance ≈ 8.9%
-
Apply the formula for average atomic mass:
- Show the calculation:
- Identify the element:
- The calculated relative atomic mass is 20.18 amu.
- Consulting the periodic table, the element with atomic mass close to 20.18 is Neon (Ne), which has a standard atomic weight of 20.180 amu.