15.2 Introduction to Proteins
1. Introduction to Proteins
Proteins are nitrogenous high molecular weight polymers. They are complex organic substances that are essential for life.
- Building Blocks: The fundamental units of all proteins are amino acids.
- Hydrolysis: Upon complete hydrolysis, proteins break down into their constituent amino acids.
- Peptide Bonds: Amino acids are linked together by peptide bonds to form polypeptide chains.
2. Classification of Proteins
Proteins can be classified based on their structure, constitution, shape, and function.
2.1. Structure-based Classification
The specific three-dimensional arrangement of a protein determines its function. Proteins are organized into four levels of structure:
-
Primary Structure:
- This refers to the unique sequence of amino acids in a polypeptide chain.
- Amino acids are linked by peptide bonds.
- Example: The specific order of amino acids in insulin.
-
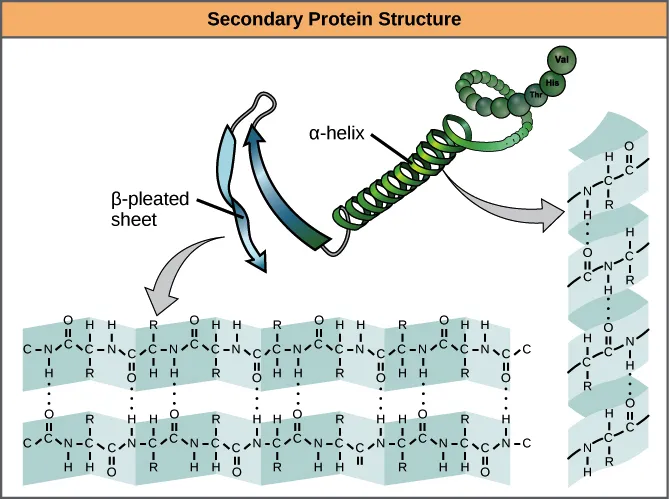
Secondary Structure:
- Describes the local spatial arrangement of the polypeptide chain, often in a spiral shape (alpha-helix) or zig-zag manner (beta-pleated sheet).
- This coiling or zig-zagging is stabilized by hydrogen bonds between the carbonyl oxygen of one peptide bond and the amide hydrogen of another.
- Example: Collagen often forms a triple helix.
-
Tertiary Structure:
- The overall three-dimensional folding of a single polypeptide chain, including its secondary structural elements.
- This unique globular shape is stabilized by various interactions such as hydrogen bonds, ionic bonds, disulfide bridges, and hydrophobic interactions.
- Example: Myoglobin, a compact globular protein.
-
Quaternary Structure:
- The arrangement of multiple folded protein subunits (polypeptide chains) in a multi-subunit complex.
- It is the highest level of protein organization.
- Stabilized by various bonding interactions including hydrogen bonding, salt bridges, and disulfide bonds.
- Example: Hemoglobin, which consists of four subunits.
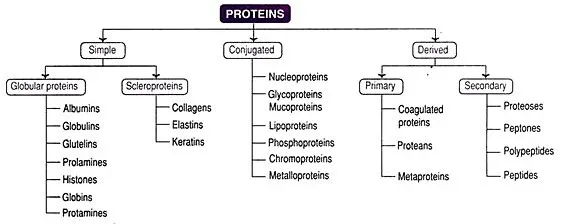
2.2. Classification based on Constitution
Proteins can be grouped by what they are made of upon hydrolysis:
-
Simple Proteins:
- Produce only amino acids upon complete hydrolysis.
- Examples: Albumins, globulins, collagens.
-
Composite or Conjugated Proteins:
- Composed of a simple protein (apoprotein) and a non-protein group (prosthetic group).
- Examples: Lipoproteins (lipid prosthetic group), glycoproteins (carbohydrate), phosphoproteins (phosphate), nucleoproteins (nucleic acid), hemoproteins (heme).
-
Derived Proteins:
- Formed from partial hydrolysis of simple or conjugated proteins by acids, bases, or enzymes.
- Examples: Peptones, peptides, proteoses, proteans.
2.3. Classification based on Shapes
Based on their overall three-dimensional shape:
| Feature | Fibrous Proteins | Globular Proteins |
|---|---|---|
| Shape | Elongated or fibrous polypeptide chains | Sphere or globe-like upon folding |
| Solubility | Generally insoluble in water | Usually soluble in water, acids, alcohol, and bases |
| Role | Structural, provide strength and support | Functional (enzymes, hormones, transport) |
| Examples | Keratins (hair, nails), Collagens, Myosins | Albumins (eggs), Hemoglobin, Enzymes, Hormones |
2.4. Classification based on Functions
Proteins perform a vast array of functions in living organisms:
- Enzymes: Catalyze biochemical reactions. (e.g., Lactase, Catalase)
- Structural Proteins: Provide support and shape to cells and tissues. (e.g., Collagen, Keratin)
- Transport Proteins: Facilitate the movement of substances. (e.g., Hemoglobin)
- Storage Proteins: Store nutrients and ions. (e.g., Ferritin, Casein)
- Hormonal Proteins: Regulate physiological processes. (e.g., Insulin)
- Defensive Proteins: Protect against pathogens. (e.g., Antibodies)
- Receptor Proteins: Bind to specific molecules and transmit signals.
- Contractile Proteins: Enable muscle contraction. (e.g., Actin and Myosin)
- Regulatory Proteins: Control protein activity or gene expression.
3. Structure-Function Relationship of Proteins
The intricate relationship between a protein's structure and its function is fundamental:
- Primary structure dictates how the protein will fold.
- Secondary structure contributes to the overall shape.
- Tertiary structure determines the active site or binding regions, crucial for function.
- Quaternary structure allows for complex regulation and cooperative binding.
Denaturation: When a protein's native structure is disrupted by heat, pH changes, or chemicals, it loses its biological activity.
4. Key Roles of Proteins in Maintaining Body Function
Proteins are indispensable macromolecules involved in virtually all biological processes.
- Structural Support: Form protoplasm and provide integrity to tissues (e.g., collagen).
- Enzymatic Activity: Act as biological catalysts.
- Hormonal Regulation: Serve as chemical messengers (e.g., insulin).
- Transport: Carry substances like oxygen (e.g., hemoglobin).
- Movement: Essential for muscle contraction (e.g., actin, myosin).
- Immune Defense: Form antibodies to identify and neutralize pathogens.
5. Fibrous Proteins for Structure and Support (Specific Examples)
Fibrous proteins are specialized for providing strength and structural support.
- Silk Fiber: Long, strong, durable, and flexible structures.
- Keratin: Found in hair and nails, providing structure and strength.
- Myosin: A muscle protein essential for contraction and relaxation.
- Fibrin: Crucial for blood clotting, forming a mesh to seal wounds.