11.5 Reactions of Acyl Chlorides
Acyl chlorides, also known as acid chlorides, are highly reactive carboxylic acid derivatives due to the strong electron-withdrawing nature of the carbonyl group and the chlorine atom, making the carbonyl carbon highly electrophilic. They readily undergo nucleophilic acyl substitution reactions.
1. Reaction of Acyl Chlorides with Alcohols (Formation of Esters)
Acyl halides react vigorously with alcohols at room temperature to produce esters and hydrogen chloride. This is a common method for synthesizing esters.
Example 1: Ethanoyl chloride with Ethanol
Ethanoyl chloride + Ethanol → Ethyl ethanoate + Hydrogen chloride
Example 2: Benzoyl chloride with Ethanol
Benzoyl chloride + Ethanol → Ethyl benzoate + Hydrogen chloride
2. Reaction of Acyl Chloride with Phenol
Acyl chlorides react with phenols at room temperature to produce esters. Phenols act as nucleophiles similar to alcohols, but typically require a base to neutralize the HCl formed, which can react with the phenol itself.
Example: Ethanoyl chloride with Phenol
Ethanoyl chloride + Phenol → Phenyl ethanoate + Hydrogen chloride
3. Reaction with Water (Hydrolysis)
Acyl halides hydrolyze readily with water at room temperature to produce carboxylic acids and hydrochloric acid. This reaction demonstrates their high reactivity towards even weak nucleophiles like water.
Example: Ethanoyl chloride with Water
Ethanoyl chloride + Water → Ethanoic acid + Hydrogen chloride
4. Reaction with Ammonia (Formation of Amides)
Acyl halides react with ammonia at room temperature to produce primary amides. The hydrogen chloride produced is acidic and reacts with excess ammonia to form ammonium chloride, an ionic salt. Therefore, at least two moles of ammonia are typically required for every mole of acyl chloride.
Reaction 1: Amide Formation
Ethanoyl chloride + Ammonia → Ethanamide + Hydrogen chloride
Reaction 2: Neutralization of HCl
Hydrogen chloride + Ammonia → Ammonium chloride
5. Reaction with Primary and Secondary Amines (Formation of N-substituted Amides)
Primary and secondary amines react in a similar fashion to ammonia at room temperature, forming N-substituted amides. The hydrogen chloride byproduct reacts with excess amine to form an ammonium salt. Tertiary amines, lacking an N-H bond, cannot form amides but can react as bases to neutralize HCl.
Example: Ethanoyl chloride with Methanamine (a primary amine)
Ethanoyl chloride + Methanamine → N-methylmethanamide + Hydrogen chloride
Similar to ammonia, the HCl produced would react with an additional mole of methanamine to form methylammonium chloride ().
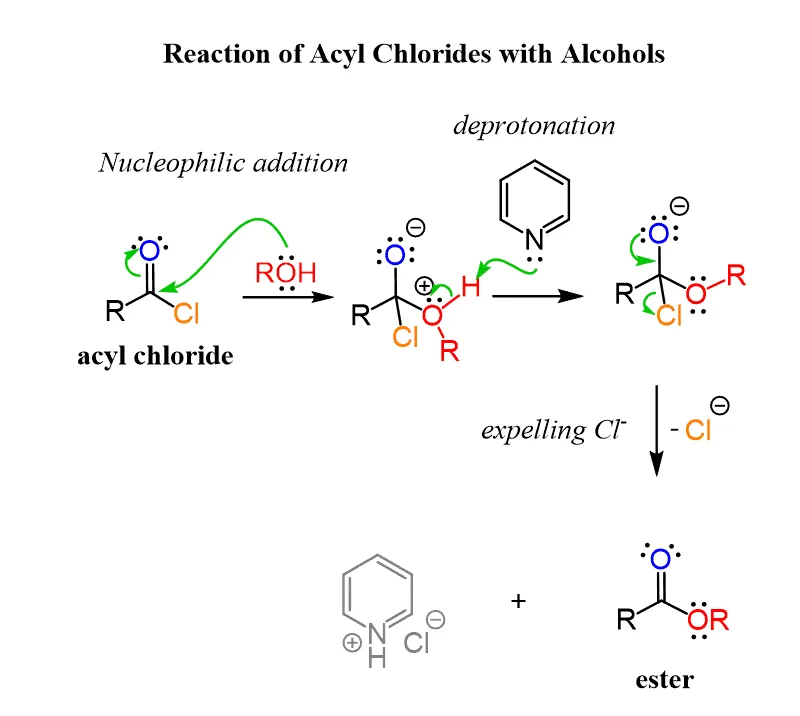
General Mechanism of Nucleophilic Acyl Substitution
The reactions of acyl chlorides with nucleophiles (alcohols, phenols, water, amines) typically proceed via a nucleophilic acyl substitution mechanism. This is an addition-elimination pathway involving a tetrahedral intermediate.
- Nucleophilic Attack: The nucleophile (e.g., alcohol, water, amine) attacks the highly electrophilic carbonyl carbon. This forms a tetrahedral intermediate.
- Elimination of Leaving Group: The chloride ion () is a good leaving group and is expelled from the tetrahedral intermediate, restoring the carbonyl group.
Here is a general representation of the mechanism:
Comparison of Hydrolysis of Acyl Chlorides, Alkyl Chlorides, and Aryl Chlorides
The ease of hydrolysis, which is a nucleophilic substitution reaction, varies significantly among these chlorinated organic compounds. The trend is:
This trend can be explained by examining the strength and polarity of the bond and the electrophilicity of the carbon atom attached to the chlorine.
-
Acyl Chlorides (e.g., ) In acyl chlorides, the carbonyl carbon atom is attached to two electronegative atoms (oxygen and chlorine), both strongly pulling electron density away. This makes the carbonyl carbon highly electron-deficient and therefore extremely susceptible to nucleophilic attack. The nucleophile can easily attack this carbon, leading to the displacement of the chlorine atom (a good leaving group). Consequently, hydrolysis occurs most readily, even with a weak nucleophile like water.
-
Alkyl Chlorides (e.g., ) In alkyl chlorides, the carbon atom attached to chlorine is bonded to only one electronegative atom (chlorine). While the carbon atom does have a partial positive charge () due to the electron-withdrawing effect of chlorine, it is less electron-deficient compared to the carbonyl carbon in acyl chlorides. Hydrolysis of alkyl chlorides typically requires a stronger nucleophile than water (e.g., ) or harsher conditions (heating, presence of a catalyst). Represented by:
-
Aryl Chlorides (e.g., Chlorobenzene, ) In aryl chlorides, the lone pair of electrons on the chlorine atom is involved in resonance with the benzene ring. This resonance delocalization imparts partial double bond character to the bond in aryl chlorides. As a result, the bond is significantly stronger and shorter than a typical single bond. Breaking this bond is very difficult, even with a strong nucleophile and harsh conditions. Therefore, aryl chlorides are generally unreactive towards hydrolysis under normal conditions.
Possible Questions and Answers
Q: What is the general type of reaction that acyl chlorides undergo with nucleophiles? A: Nucleophilic acyl substitution, specifically an addition-elimination mechanism involving a tetrahedral intermediate.
Q: Name the product formed when ethanoyl chloride reacts with excess ethylamine (). A: N-ethylethanamide () and ethylammonium chloride ().
Q: Explain why acyl chlorides are much more reactive towards hydrolysis than alkyl chlorides. A: In acyl chlorides, the carbonyl carbon is bonded to two electronegative atoms (O and Cl), making it highly electron-deficient and very susceptible to nucleophilic attack. In alkyl chlorides, the carbon is only bonded to one electronegative atom (Cl), making it less electron-deficient and thus less reactive to nucleophiles.
Q: Why are aryl chlorides unreactive towards hydrolysis? A: The chlorine atom's lone pair participates in resonance with the aromatic ring, giving the C-Cl bond partial double bond character. This makes the bond stronger and harder to break via nucleophilic substitution.