9.2 Reactivity of Halogenoalkanes
The reactivity of halogenoalkanes (also known as alkyl halides) and aryl halides is primarily determined by the strength and polarity of the carbon-halogen (C-X) bond.
C-X Bond Polarity
Halogens are more electronegative than carbon. This difference in electronegativity causes the C-X bond to be polarized. The carbon atom attached to the halogen acquires a partial positive charge (), making it electrophilic, while the halogen atom acquires a partial negative charge (), making it nucleophilic.
This polarization makes the carbon atom susceptible to attack by nucleophiles. For example, chloroethane can undergo a nucleophilic substitution reaction where the chlorine atom is replaced by another nucleophile ():
Comparison: Alkyl Halides vs. Aryl Halides
The reactivity differs significantly between alkyl halides and aryl halides (e.g., chlorobenzene).
- Alkyl Halides: The C-X bond is polarized, and the halogen can be readily replaced by a nucleophile via nucleophilic substitution.
- Aryl Halides (e.g., Chlorobenzene):
- The lone pair of electrons on the chlorine atom is delocalized over the benzene ring through resonance.
- This delocalization gives the C-Cl bond a partial double bond character, making it stronger and more difficult to break compared to an alkyl C-Cl bond.
- Consequently, the chlorine atom in chlorobenzene cannot be easily replaced by a nucleophile, meaning it does not readily undergo nucleophilic substitution.
- Instead, the chlorine atom increases the electron density of the benzene ring due to resonance, directing incoming electrophiles to the ortho and para positions. Thus, chlorobenzene primarily undergoes electrophilic substitution reactions.
For instance, nitration of chlorobenzene yields a mixture of o-nitrochlorobenzene and p-nitrochlorobenzene:
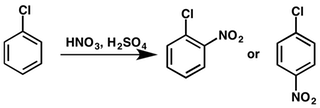
Reactions of Halogenoalkanes or Alkyl Halides
As discussed, the polar character of the C-X bond, known as the inductive effect, is responsible for the characteristic chemical reactions of alkyl halides.
Alkyl halides primarily undergo two types of reactions:
- Nucleophilic substitution reactions ( Reactions)
- Elimination reactions
Nucleophilic Substitution () Reactions of Alkyl Halides
A nucleophilic substitution reaction is one where a halogen atom of an alkyl halide is substituted or replaced by a strong nucleophile. These reactions involve three key components: the Substrate, the Nucleophile, and the Leaving Group.
- Substrate Molecule: This is the alkyl halide molecule upon which the nucleophile attacks.
- Nucleophile ( or ): A species possessing a lone pair of electrons, which it donates to the electrophilic carbon of the alkyl halide. Nucleophiles can be neutral or negatively charged.
Examples of Nucleophiles
| Nucleophile | Name |
|---|---|
| Hydroxide ion | |
| Ethoxide ion | |
| Hydrogen sulfide ion | |
| Thiocyanate ion | |
| Amino group | |
| Chloride ion | |
| Bromide ion | |
| Ammonia | |
| Water |
- Leaving Group (LG): The halogen atom of the alkyl halide is the leaving group. It departs with an unshared pair of electrons and acts as a nucleophile itself. For a successful substitution, the incoming nucleophile must generally be stronger than the departing leaving group.
- Good leaving groups: , , ,
- Poor leaving groups: , ,
- Note: Iodide ion () is both a good nucleophile and a good leaving group.
There are two fundamental events in a nucleophilic substitution reaction:
- Breaking of the bond between carbon and halogen.
- Formation of a new bond between carbon and the attacking nucleophile.
The specific mechanism of substitution is significantly influenced by the nature of the attacking nucleophile, the substrate (alkyl halide), and the leaving group. This leads to two distinct mechanisms: and .
Carbocations and their Stability
A carbocation is a carbon atom that is attached to three atoms or groups and possesses a unit positive charge. Carbocations are often intermediates in reactions.
The general stability order of simple alkyl carbocations is:
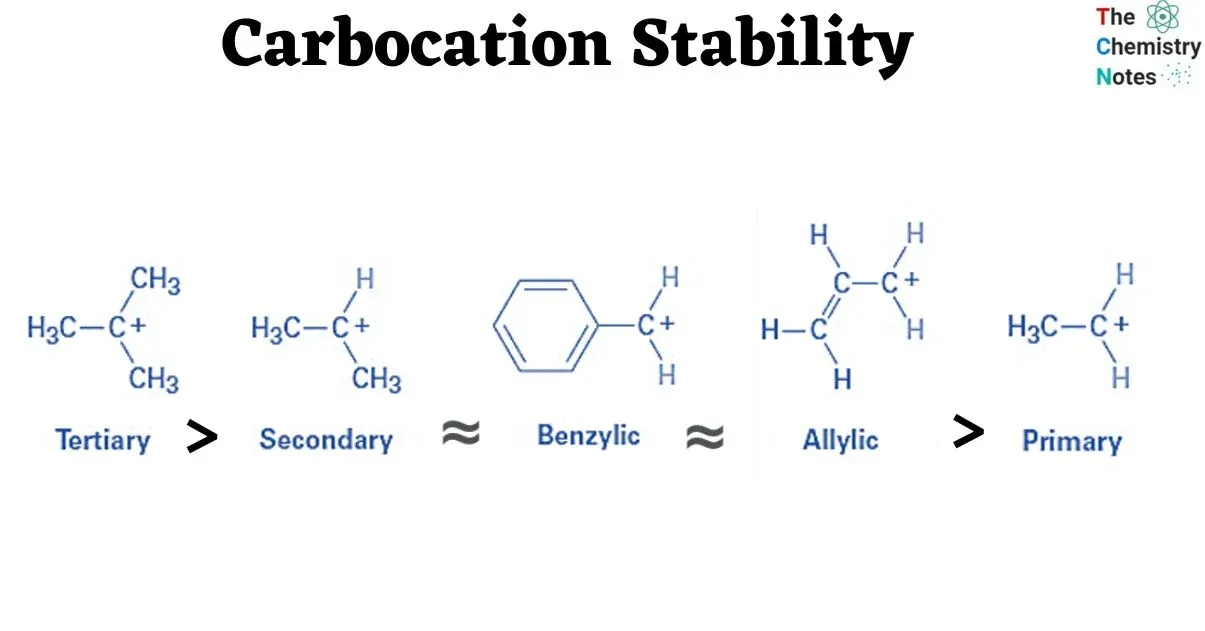
This order is observed because alkyl groups are weak electron-donating groups due to hyperconjugation and inductive effects. The positive charge on the carbon is delocalized and stabilized by the electron donation from the surrounding alkyl groups. Therefore, the stability of a carbocation increases with an increase in the number of alkyl groups attached to the positively charged carbon.
Reaction with Aqueous Silver Nitrate
When a haloalkane (alkyl halide) reacts with aqueous silver nitrate (), the silver ion () in the solution undergoes an exchange reaction with the halide ion () from the haloalkane. This results in the formation of a silver halide precipitate ().
Where X represents a halogen: Cl, Br, or I.
Note: Alkyl fluorides (R-F) do not react readily with because the C-F bond is exceptionally strong and the fluoride ion is not easily displaced.
The color of the silver halide precipitate depends on the specific halogen present:
- AgCl: White precipitate, soluble in (aqueous ammonia) solution.
- AgBr: Cream-colored precipitate, partially soluble in solution.
- AgI: Yellow precipitate, insoluble in solution.