7.1.2 Optical Isomerism and Chirality in Organic Chemistry
Introduction to Chirality
Chirality describes objects that are non-superimposable on their mirror images. A common analogy is our hands: the right hand is a mirror image of the left hand, but no matter how we orient them, they cannot perfectly overlap.
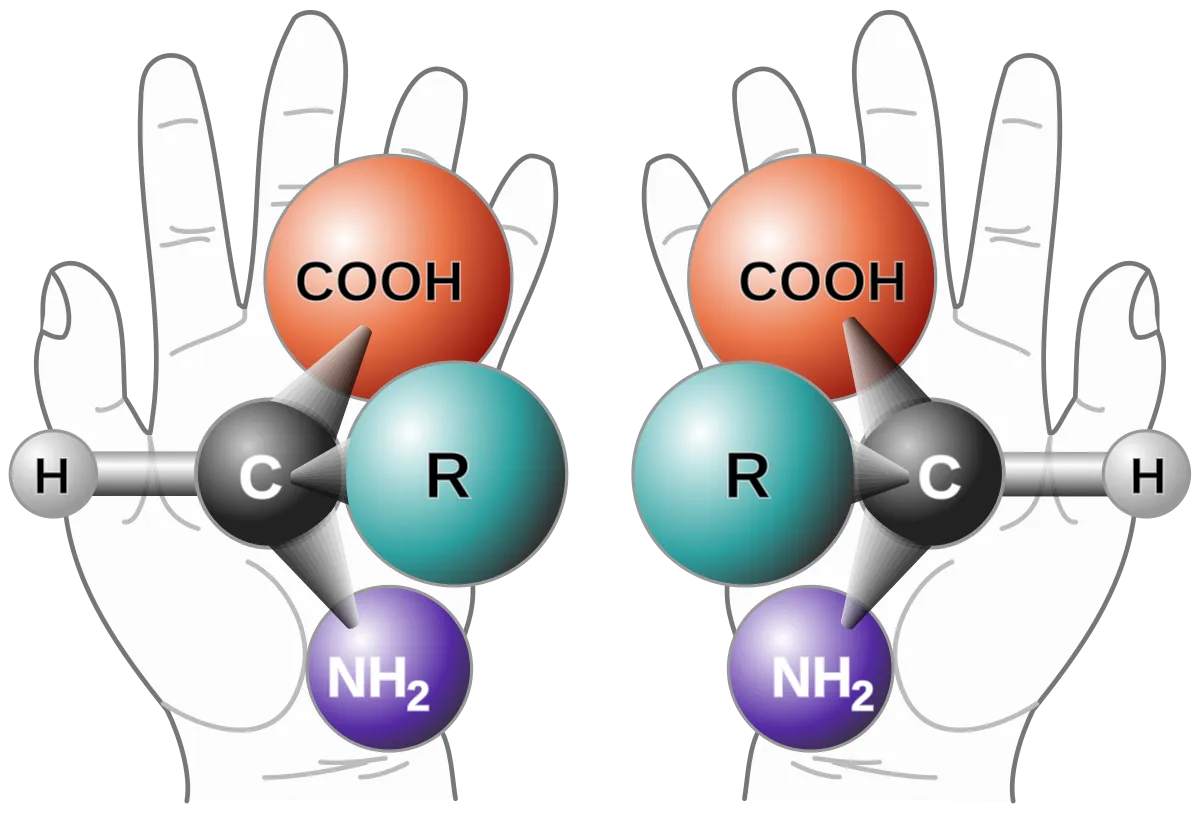
- Chiral Objects: Objects that possess "handedness" and are non-superimposable on their mirror images. Examples include hands, scissors, keyboards, and many organic molecules. The Greek word "cheir" means 'hand'.
- Achiral Objects: Objects that are superimposable on their mirror images (they lack handedness).
Chiral Molecules and Asymmetric Carbons
Many organic molecules are chiral. A molecule is chiral if it contains at least one chiral carbon (also known as an asymmetric carbon or stereocenter).
A chiral carbon is a carbon atom bonded to four different atoms or groups of atoms.
In contrast, achiral molecules have a central carbon atom bonded to identical groups, making their mirror images superimposable and thus identical.
Example: Bromochlorofluoromethane () is a chiral molecule because its central carbon is bonded to four different groups: hydrogen (), bromine (), chlorine (), and fluorine (). Its mirror image is non-superimposable.
Enantiomers and Enantiomerism
When two stereoisomers are non-superimposable mirror images of one another, they are called enantiomers. The phenomenon is known as enantiomerism. The word "enantiomer" comes from the Greek "enantion", meaning 'opposite'.
- Enantiomers have identical physical properties (boiling point, melting point, density) except for their interaction with plane-polarized light.
- They also have identical chemical properties in an achiral environment but react differently in a chiral environment or with other chiral molecules.
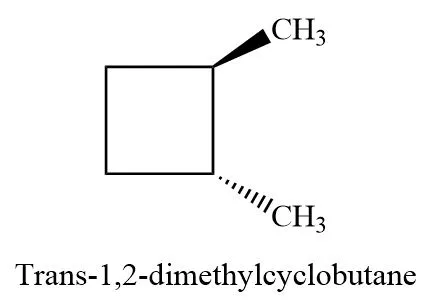
Example: The two mirror image forms of trans-1,2-dimethylcyclobutane are enantiomers.
Optical Isomerism and Plane-Polarized Light
-
Ordinary Light: Consists of waves oscillating in all planes perpendicular to its direction of propagation.
-
Plane-Polarized Light: When ordinary light passes through a polarizing filter (calcite or Polaroid filter), only light waves vibrating in a single parallel plane are allowed to pass.
-
Optical Isomers: Isomers that can rotate the plane of plane-polarized light either clockwise or anticlockwise. This phenomenon is called optical isomerism.
-
Dextrorotatory (d or +): Rotates plane-polarized light in a clockwise direction.
-
Levorotatory (l or -): Rotates plane-polarized light in an anticlockwise direction.
The symbols "d" and "l" are now largely obsolete; IUPAC recommends "+" and "-" symbols for dextrorotatory and levorotatory isomers, respectively.
Racemic Mixtures
A racemic mixture (or racemate) is a 50-50 mixture of two enantiomers.
A racemic mixture is optically inactive. This is because the two enantiomers rotate plane-polarized light to an equal extent but in opposite directions, effectively canceling out each other's optical activity.
Significance of Chirality in Biology and Pharmaceuticals
Chirality is highly significant in biological systems and pharmaceutical chemistry. Biological receptors (enzymes, antibodies) are chiral, and thus they often interact differently with the two enantiomers of a chiral molecule.
Examples:
- Nicotine: The nicotine molecules naturally synthesized by the tobacco plant are chiral and exhibit optical isomerism.
- Ibuprofen: A common painkiller, ibuprofen, is a chiral molecule. One enantiomer provides the intended therapeutic effect, while the other is inactive or can even have damaging effects.
Worked Examples
Concept Assessment 7.2: Separate chiral and achiral molecules in the following.
To determine if a molecule is chiral, identify if it contains a carbon atom bonded to four different groups.
a.
- Analyze Carbon 1 (): Bonded to three Fluorine atoms. Not chiral.
- Analyze Carbon 2 (): Bonded to two Hydrogen atoms. Not chiral.
- Analyze Carbon 3 (): Bonded to three Chlorine atoms. Not chiral.
- Result: Achiral
b.
- Analyze Carbon 2 (): Bonded to one Hydrogen (), one Fluorine (), one group, and one group. All four groups are different.
- Result: Chiral
c.
- Analyze Carbon at : Bonded to , , , and . All four groups are different.
- Result: Chiral
d.
- Analyze Carbon at : Bonded to , , , and ethyl group. All four groups are different.
- Result: Chiral
e.
- Analyze Carbon at : Bonded to , , , and . All four groups are different.
- Result: Chiral
Possible Questions and Answers
Q: What is a chiral carbon? A: A carbon atom bonded to four different atoms or groups of atoms.
Q: Distinguish between chiral and achiral objects. A: Chiral objects are non-superimposable on their mirror images (hands), while achiral objects are superimposable on their mirror images (a cup).
Q: Define enantiomers. A: Enantiomers are stereoisomers that are non-superimposable mirror images of each other.
Q: Why is a racemic mixture optically inactive? A: A racemic mixture contains a 50-50 ratio of two enantiomers. Since enantiomers rotate plane-polarized light to an equal extent but in opposite directions, their effects cancel out, rendering the mixture optically inactive.
Q: What is plane-polarized light? A: Light in which waves oscillate in only one plane, typically produced by passing ordinary light through a polarizing filter.