6.6 Stereoisomerism in Coordination Compounds
This section explores stereoisomerism in coordination compounds, a phenomenon where compounds have the same chemical formula and connectivity but differ in the spatial arrangement of their atoms or ligands. Two types are examined: geometric isomerism and optical isomerism.
Introduction to Stereoisomerism
Stereoisomerism in coordination compounds arises from different arrangements of atoms or ligands of complexes in space. These isomers have identical chemical formulas but distinct three-dimensional structures. Stereoisomers are broadly categorized into:
- Geometric isomers (also known as cis-trans isomers or diastereomers)
- Optical isomers (also known as enantiomers)
i. Geometric Isomerism
Geometric isomerism is observed in complexes where ligands are arranged differently in space relative to the central metal ion. This leads to distinct spatial configurations, often termed cis and trans.
Conditions for Geometric Isomerism
Geometric isomerism is characteristic of complexes with coordination number 4 (square planar) and 6 (octahedral).
Tetrahedral complexes cannot show geometric isomerism because all four ligands are equidistant and adjacent to each other, making all positions equivalent.
Examples of Geometric Isomerism
1. Square Planar Complexes
Consider diamminedichloroplatinum(II), , which is a square planar complex.
- Cis-isomer: The identical ligands (e.g., both groups or both ligands) are positioned next to each other.
- Trans-isomer: The identical ligands are positioned opposite to each other.
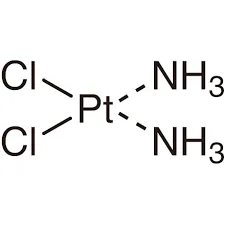
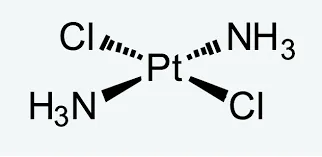
These isomers of platinum have remarkably different biological behaviors.
2. Octahedral Complexes
Octahedral complexes, such as tetraamminediaquacobalt(III) ion, , also exhibit geometric isomerism.
- Cis-isomer: The two water ligands (or four ammonia ligands) are next to each other.
- Trans-isomer: The two water ligands (or four ammonia ligands) are opposite to one another.
3. Octahedral Complexes with Bidentate Ligands
Bis(1,2-diaminoethane)diaquanickel(II) ion, , also exhibits geometric isomerism.
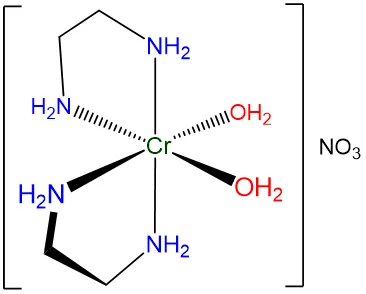
Properties of Geometric Isomers: Polarity and Symmetry
Geometric isomers exhibit different properties due to their distinct symmetry and polarity.
Cis-isomers:
- Generally polar because they possess an imbalance of electron cloud distribution and lack overall symmetry.
- For example, in a complex with ligands like and , the higher electronegativity of oxygen in water compared to nitrogen in ammonia causes electron density to shift towards the water ligands, creating individual dipoles that sum up to a net dipole moment for the complex.
Trans-isomers:
- Generally non-polar due to their symmetric shapes and even distribution of the electron cloud.
- The individual bond dipoles generated by opposing ligands cancel each other out, resulting in a zero or negligible net dipole moment.
Fighting Cancer: Cisplatin
- Discovery: In 1965, Barnett Rosenberg discovered that cis-platin, , possessed the property of inhibiting cell division and could be used as an anticancer drug. Trans-platin, however, had no such medicinal properties.
- Mechanism of Action:
- Cis-platin passes through cell membranes into the nucleus of cancerous cells.
- Inside the nucleus, the donor atoms of the DNA strand replace the chloride ligands and bind strongly to the platinum atoms, forming a bridge.
- This disrupts the normal replication process of DNA, causing cancerous cells to die.
- Side Effects: It affects healthy fast-growing cells, leading to side effects like hair loss and kidney damage.
ii. Optical Isomerism
Optical isomers are molecules that are non-superimposable mirror images of each other. They are also referred to as enantiomers.
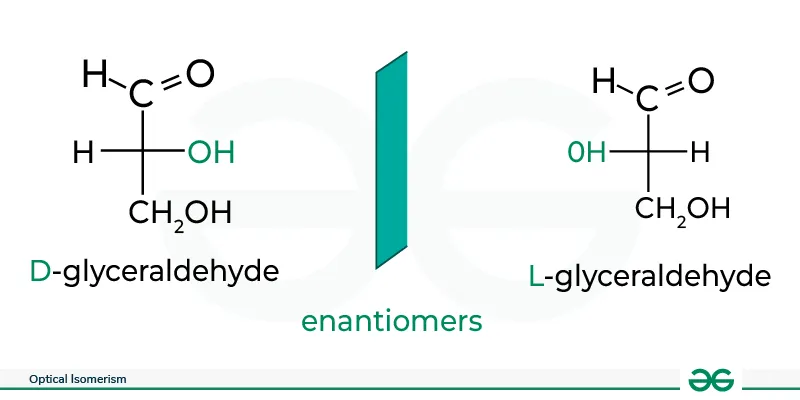
Properties of Optical Isomers
- Optical isomers have identical physical properties (melting point, boiling point, etc.) except for their interaction with plane-polarized light.
- They rotate the plane of polarized light by the same magnitude but in opposite directions (clockwise vs. counterclockwise).
Conditions for Optical Isomerism
- Optical isomerism is typically exhibited by octahedral complexes that lack a plane of symmetry.
- In octahedral complexes with two monodentate ligands and two bidentate ligands (e.g., ), only the cis-isomer can show optical isomerism. The trans-isomer has a plane of symmetry and is optically inactive.
- Octahedral complexes containing three bidentate ligands (e.g., like ) are inherently chiral and exhibit optical isomerism.