5.6 Introduction: General Tendency to Form Complexes
Group 2 elements, also known as the alkaline earth metals, almost exclusively form ionic compounds containing the ion. Compared to the alkali metals (Group 1), they exhibit a significantly greater tendency to form complexes with Lewis bases.
The factors contributing to this greater tendency are:
- Higher Positive Charge: The ions have a charge, which is twice that of alkali metal ions ().
- Smaller Ionic Radii: For a given period, alkaline earth metal ions have smaller ionic radii than alkali metal ions.
- High Charge Density: The combination of higher charge and smaller radii results in a high charge density, making them stronger Lewis acids and enhancing their ability to attract and coordinate with Lewis bases.
This tendency to form complexes is most pronounced for the lightest cation, , and decreases rapidly as the radius of the metal ion increases down the group ().
Beryllium Complexes ()
The chemistry of the ion is largely dominated by its behavior as a strong Lewis acid, readily forming complexes with various Lewis bases. Beryllium achieves an octet of electrons around itself in these complexes.
1. Hydrated Beryllium Ion and its Acidity
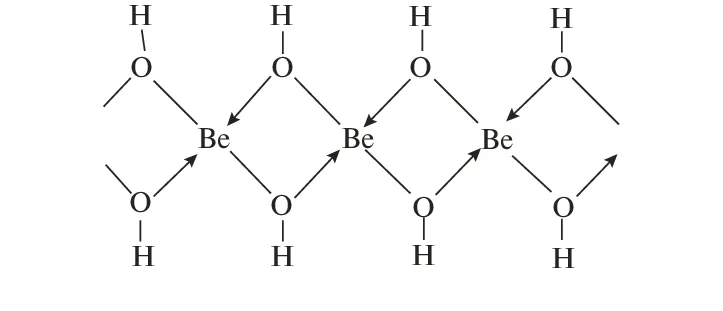
Formation: salts dissolve in water to form acidic solutions. This is due to the formation of the tetrahedral aquo complex, . In this complex, the beryllium ion is coordinated to four water molecules.
- Coordination Number: 4
- Geometry: Tetrahedral
Acidity: Due to its exceptionally high charge-to-radius ratio, the ion strongly polarizes the coordinated water molecules. This polarization weakens the bonds in the water molecules, making the protons more acidic and easily removed by a solvent water molecule.
The acidity can be represented by the following equilibrium:
2. Other Beryllium Complexes
Hydroxo Complexes: In the presence of a strong base, beryllium and its salts form the tetrahedral hydroxo complex, . This explains why beryllium oxide () is amphoteric, reacting with both acids and strong bases.
Fluoride Complexes: Beryllium forms very stable tetrahedral fluoride complexes, such as and . This stability is again attributed to the small size and high charge density of the ion, allowing for strong electrostatic interactions with fluoride ions.
Lewis Acid Behavior of Halides: Beryllium halides (e.g., ) behave like Lewis acids by readily forming adducts with various Lewis bases.
Visualizing Beryllium Complex Formation
The following visual aid illustrates the electronic configuration changes leading to the formation of :
Complexes of Heavier Alkaline Earth Metals ( etc.)
The heavier alkaline earth metal ions also form complexes, but generally to a lesser extent than . Their larger ionic radii result in lower charge densities, making them weaker Lewis acids.
- Coordination Number: These ions typically exhibit higher coordination numbers, often 6 or more, reflecting their larger size which can accommodate more ligands around them.
- Significance: Complex formation is still important for the smaller cations among the heavier group, particularly and .
Example: Hydrated Magnesium Ion: Aqueous solutions of contain the octahedral aquo complex, .
- Coordination Number: 6
- Geometry: Octahedral
- Orbital Involvement: In this complex, can extend its coordination number to six by utilizing one , three , and two orbitals present in its outermost shell to accommodate the six lone pairs from water molecules.
Biological and Analytical Importance
Complexes of Magnesium and Calcium are vital in biological systems and analytical chemistry.
- Chlorophyll: Magnesium is the central metal ion in chlorophyll, coordinated to a porphyrin ring system.
- EDTA Complexes: and form stable complexes with EDTA (Ethylenediaminetetraacetic acid). This is used to determine water hardness.