5.1 Group 2 Elements: General Trends in Physical Properties and Chemical Reactivity
The Group 2 elements, also known as alkaline earth metals, exhibit clear periodic trends in their physical and chemical properties. This section explores these trends, focusing on ionization energy, atomic radius, melting point, and overall chemical reactivity.
General Trends of Physical Properties and Chemical Reactivity
All Group 2 metals are characterized by their ability to donate two outermost electrons to form ions with a +2 charge. This electron donation process means they are oxidized, and thus they act as reducing agents. As one descends Group 2, the metals generally become more reactive.
Oxidizing And Reducing Agents→
Trends in Ionization Energy and Chemical Reactivity
Ionization energy is defined as the minimum amount of energy required to remove one mole of electron from one mole of isolated gaseous atoms or ions.
- Trend Down the Group: Both the first and second ionization energies decrease as you go down Group 2. This is illustrated in Figure 5.1.
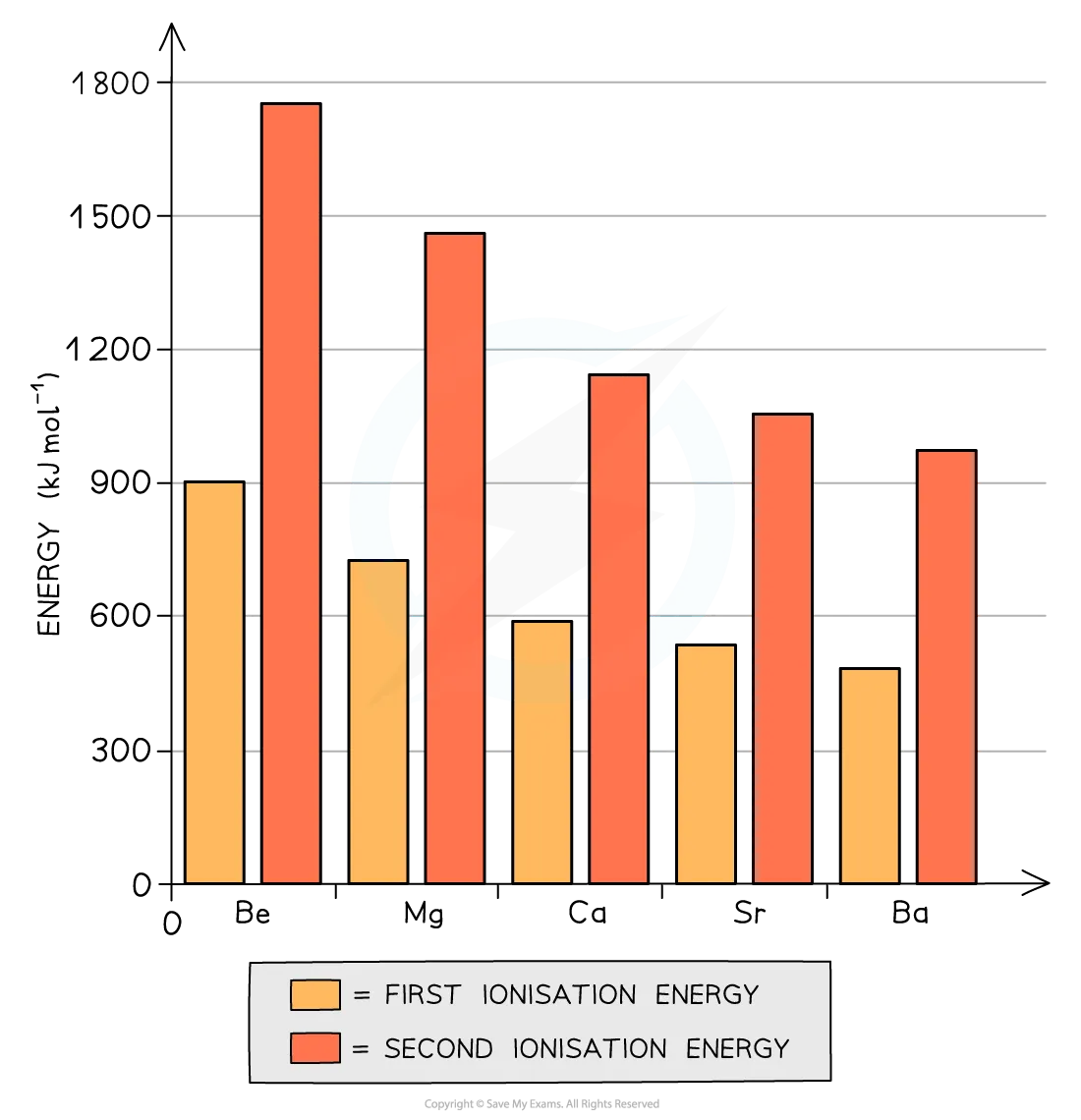
The decreasing order of first ionization energy for Group 2 elements is:
- Explanation: As we move down the group, the atoms have more electron shells, leading to:
- Increased Shielding Effect: Inner electrons shield the outermost electrons from the nucleus positive charge more effectively.
- Larger Distance: The outermost electrons are further away from the nucleus.
These factors reduce the attraction between the nucleus and the valence electrons, making it easier to remove the outer two electrons.
-
Impact on Reactivity: Since it becomes easier for atoms to lose two electrons and form 2+ ions down the group, the elements become more reactive. This trend is evident in their reactions with dilute hydrochloric acid and oxygen.
-
With dilute hydrochloric acid: The reactions become more vigorous, with bubbles of hydrogen gas being given off much faster. For example:
-
With oxygen: Metals become more reactive with oxygen down the group. Barium (Ba) is so reactive that it must be stored under oil to prevent it from reacting with atmospheric oxygen.
-
Trends in Atomic Radius
- Trend Down the Group: The atomic radii of Group 2 elements increase as you go down the group. This trend is shown in Figure 5.2.
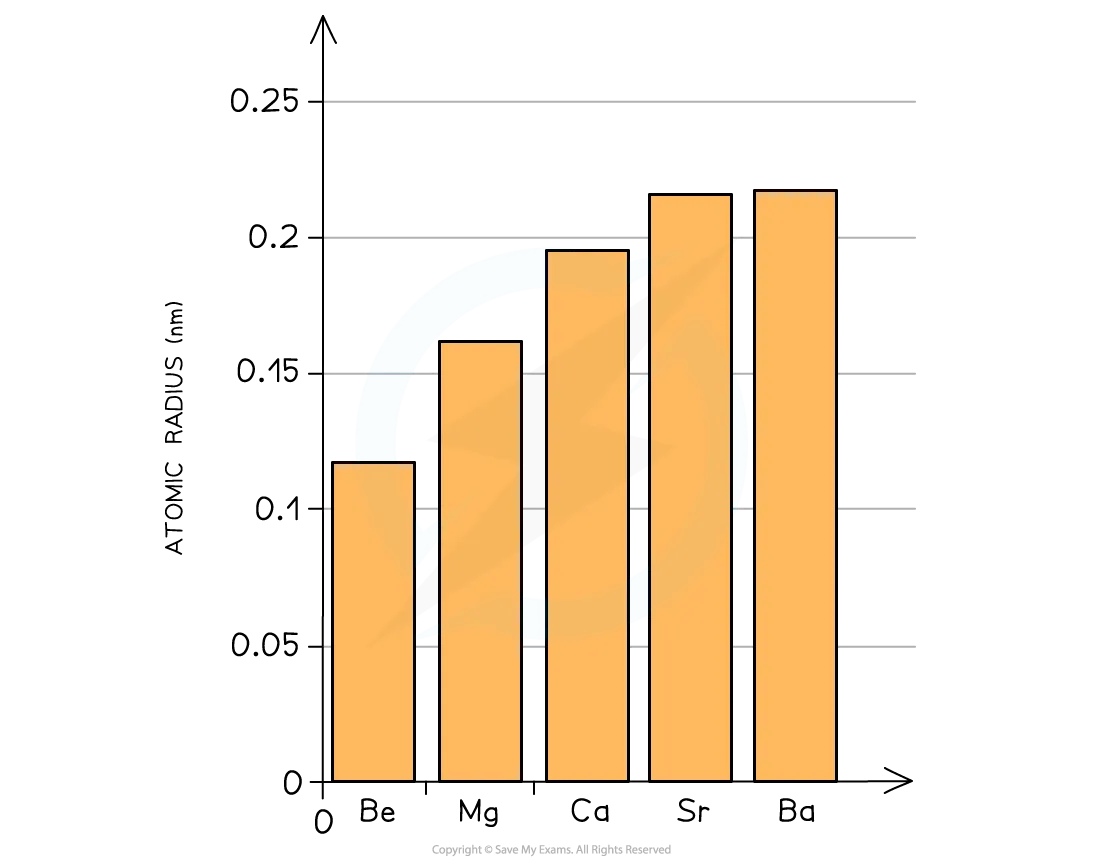
The increasing order of atomic radius for Group 2 elements is:
- Explanation: This increase in size is primarily due to:
- Increase in Extra Shells: Each successive element down the group adds a new principal electron shell.
- Shielding Effect: The increased number of inner electron shells enhances the shielding effect, reducing the effective nuclear charge felt by the valence electrons.
Trends in Melting Point
- Trend Down the Group: The melting point of Group 2 elements generally decreases as you go down the group (though there can be minor irregularities). Figure 5.3 illustrates this trend.

- Explanation: Group 2 metals form metallic bonds. As the outer electrons get further away from the nucleus (due to increasing atomic size), the attraction between the positively charged metal ions and the delocalized valence electrons decreases. This results in a weaker metallic bond, which requires less energy to overcome, thus causing a decrease in the melting point.
Summary of Physical Properties for Alkaline Earth Metals
Table 5.1 provides a summary of the general trends in physical properties such as atomic radius, melting point, and density for Group 2 elements.
Table 5.1: Physical properties of alkaline earth metals
| Group 2 Element | Atomic radius/nm | Atomic number | Melting point/°C | Density/g cm |
|---|---|---|---|---|
| Beryllium (Be) | 0.122 | 4 | 1280 | 1.85 |
| Magnesium (Mg) | 0.160 | 12 | 650 | 1.74 |
| Calcium (Ca) | 0.197 | 20 | 838 | 1.55 |
| Strontium (Sr) | 0.215 | 38 | 768 | 2.66 |
| Barium (Ba) | 0.217 | 56 | 714 | 3.56 |
Possible Questions and Answers
-
Q: Why do Group 2 elements act as reducing agents? A: Group 2 elements readily lose their two outermost electrons to form stable 2+ ions, undergoing oxidation in the process. This ability to donate electrons makes them effective reducing agents.
-
Q: Explain the trend in atomic radius down Group 2. A: Atomic radius increases down Group 2 because each successive element adds an additional electron shell. This increases the distance of the valence electrons from the nucleus and enhances the shielding effect from inner electrons, reducing the nuclear attraction and expanding the electron cloud.
-
Q: How does the reactivity of Group 2 metals change down the group, and what causes this change? A: Reactivity increases down Group 2. This is due to the decrease in ionization energy. As atoms get larger and shielding increases, it becomes easier to remove the two valence electrons, making the elements more likely to react and form 2+ ions.
-
Q: Write the balanced chemical equation for the reaction of calcium with dilute hydrochloric acid. A: