4.4 pH Titration Curves
This section explores pH titration curves, which are graphical representations of pH change during an acid-base titration. These curves are essential for understanding acid and base properties and determining unknown solution concentrations.
Introduction to Titration
Titration is a quantitative analytical technique used in neutralization reactions to determine the unknown concentration of an analyte using a titrant of known concentration.
- Titrant: The solution of known concentration added from a burette.
- Analyte: The solution of unknown concentration in the conical flask.
- Indicator: A substance added to the analyte that changes color at the endpoint of the titration.
- Endpoint: The point in a titration where the indicator changes color, signaling the completion of the reaction as observed visually.
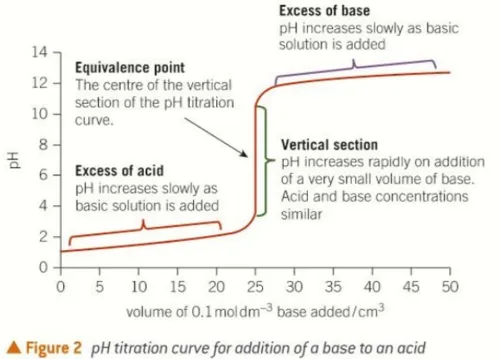
- Equivalence Point: The theoretical point where the number of moles of titrant added exactly equals the number of moles of analyte, resulting in complete neutralization. This is the midpoint of the vertical region of a titration curve.
Mathematically, at the equivalence point:
The pH at the equivalence point varies depending on the strengths of the acid and alkali involved.
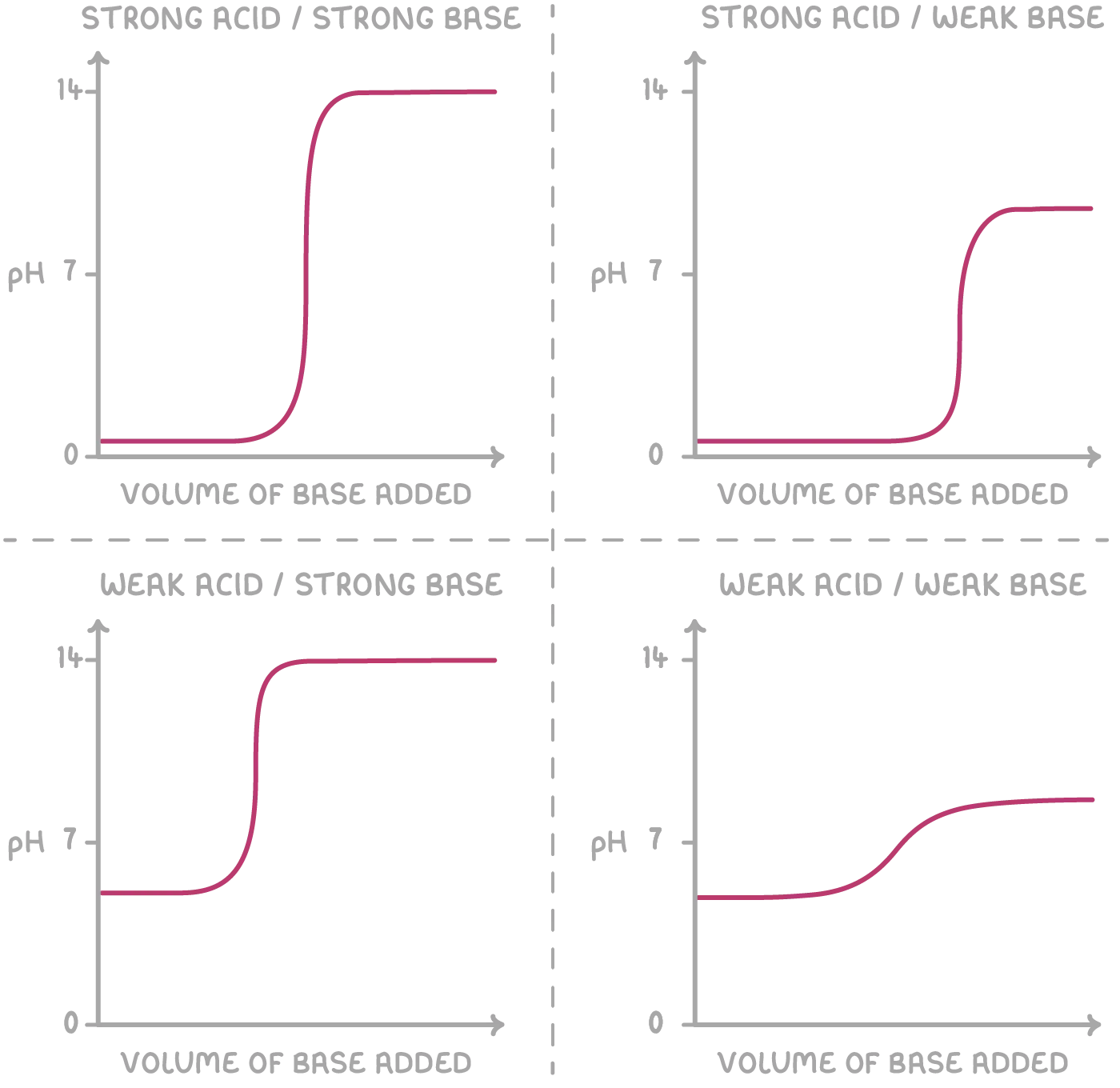
Types of pH Titration Curves
The shape of a pH titration curve depends on the strengths of the acid and base being titrated. The four main types are analyzed below.
a. Strong Acid and Strong Alkali pH Titration Curve
This type involves a strong acid (e.g., HCl) and a strong alkali (e.g., NaOH).
-
Scenario: Strong Acid (analyte) in flask, Strong Alkali (titrant) in burette
- Initial pH: Starts very low (pH 1-2) due to the presence of a strong acid ( ions).
- pH Change: As the strong alkali (NaOH) is added, the pH increases gradually as ions react with ions to form water. The pH remains relatively low until close to the equivalence point.
- Vertical Region: A very steep rise in pH occurs around the equivalence point.
- Equivalence Point: The pH is 7 (neutral) because all ions from the strong acid have been neutralized by ions from the strong base, forming a neutral salt (e.g., NaCl) and water.
- Final pH: Increases sharply to a high pH (pH 13-14) as excess strong alkali is added.
- Scenario: Strong Alkali (analyte) in flask, Strong Acid (titrant) in burette
- The curve has a similar shape but is inverted. It starts at a high pH ( 13-14) and decreases.
- The equivalence point is still at pH = 7.
- The final pH is very low ( 1-2).
b. Strong Acid and Weak Alkali pH Titration Curve
This titration involves a strong acid (e.g., HCl) and a weak alkali (e.g., ).
-
Scenario: Strong Acid (analyte) in flask, Weak Alkali (titrant) in burette
- Initial pH: Very low (pH 1-2) due to the strong acid.
- pH Change: The pH increases gradually as the weak alkali is added, reacting with ions. There is a buffering region where the solution contains both the weak base () and its conjugate acid ().
- Vertical Region: A less steep rise in pH compared to strong acid-strong alkali titrations.
- Equivalence Point: The pH is acidic (pH 5.5). This is because the salt formed () contains the ammonium ion (), which is the conjugate acid of the weak base. hydrolyzes water to produce ions.
- Final pH: Increases, but typically remains below that of a strong alkali (e.g., pH 9-11) as excess weak alkali is added.
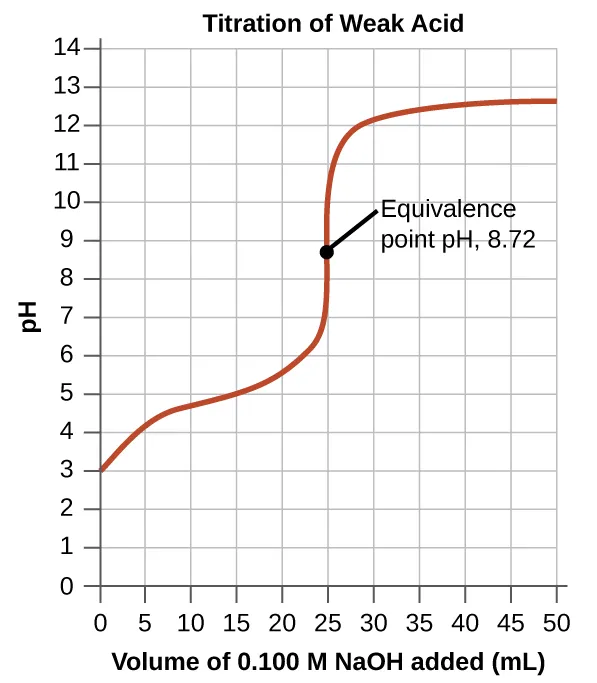
c. Weak Acid and Strong Alkali pH Titration Curve
This titration involves a weak acid (e.g., ) and a strong alkali (e.g., NaOH).
-
Scenario: Weak Acid (analyte) in flask, Strong Alkali (titrant) in burette
- Initial pH: Moderately low (pH 2-3) due to the presence of a weak acid.
- pH Change: Initially, there is a relatively flat region known as the buffer region. Here, the solution contains a significant amount of the weak acid and its conjugate base (), resisting changes in pH.
- Vertical Region: A steep rise in pH occurs around the equivalence point.
- Equivalence Point: The pH is basic (pH 9). This is because the salt formed () contains the ethanoate ion (), which is the conjugate base of the weak acid. hydrolyzes water to produce ions.
d. Weak Acid and Weak Alkali pH Titration Curve
This titration involves a weak acid (e.g., ) and a weak alkali (e.g., ).
- Scenario: Weak Acid (analyte) in flask, Weak Alkali (titrant) in burette
- Initial pH: Moderately low (pH 2-3) due to the weak acid.
- pH Change: The pH changes very gradually throughout the titration. There is no sharp vertical region.
- Equivalence Point: There is a "point of inflexion" rather than a steep rise. The pH at the equivalence point is difficult to determine precisely and is usually close to 7, but depends on the relative strengths ( and ) of the weak acid and weak base.
- Challenge: Due to the lack of a sharp pH change, indicators are generally not effective for this type of titration.
Worked Examples
Concept Assessment Exercise 4.3 (Problem 1)
Problem: In a titration, it is found that 25 cm³ of 0.1 M solution of NaOH is neutralised with 19 cm³ of HCl of unknown concentration. Calculate the concentration of the given HCl solution.
Reaction:
Solution:
-
Write the given values:
- Volume of NaOH () = 25 cm³
- Concentration of NaOH () = 0.1 M
- Volume of HCl () = 19 cm³
- Concentration of HCl () = unknown
-
Determine the stoichiometry: From the balanced chemical equation, the mole ratio of HCl to NaOH is 1:1.
-
Apply the formula:
-
Calculation:
Possible Questions/Answers
-
Q: What is the main difference between the endpoint and the equivalence point in a titration? A: The endpoint is the observed point where the indicator changes color, while the equivalence point is the theoretical point where the moles of acid and base are chemically equivalent.
-
Q: Why is the equivalence point for a titration of a weak acid with a strong base found at a pH greater than 7? A: At the equivalence point, the salt formed contains a conjugate base (e.g., ) which undergoes hydrolysis to produce ions, making the solution basic.
-
Q: Describe the shape of a pH titration curve when 20 cm³ of HCl (strong acid) is added to 20 cm³ of aqueous ammonia (weak alkali). A: The curve starts at a moderately high pH ( 11), decreases gradually through a buffer region, shows a vertical drop around an acidic equivalence point (pH 5.5), and levels off at a very low pH ( 1-2).
Summary
| Titration Type | Initial pH (Analyte) | Equivalence Point pH | Vertical Region | Example |
|---|---|---|---|---|
| Strong Acid - Strong Base | Very low (1-2) | 7 (Neutral) | Very steep | HCl + NaOH |
| Strong Acid - Weak Base | Very low (1-2) | < 7 (Acidic) | Less steep | HCl + |
| Weak Acid - Strong Base | Moderately low (2-3) | > 7 (Basic) | Steep | + NaOH |
| Weak Acid - Weak Base | Moderately low (2-3) | 7 (varies) | No sharp region | + |