4.2 The pH Scale
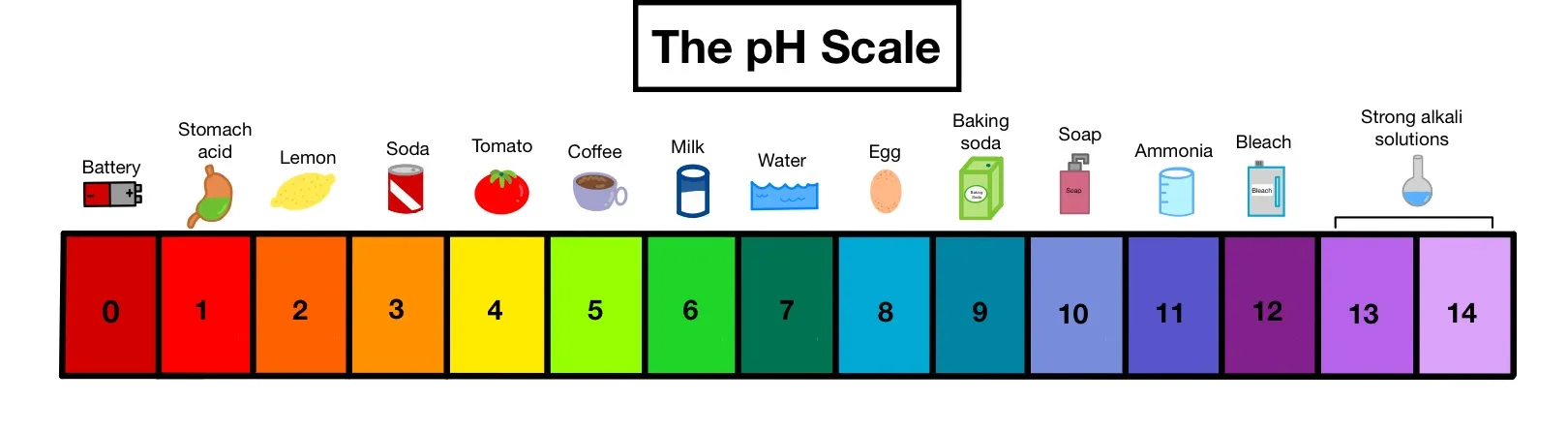
The pH scale is a fundamental concept in chemistry used to express the acidity or alkalinity of an aqueous solution. It provides a numerical measure that indicates the concentration of hydrogen ions () in a solution.
Understanding the pH Scale
- Range: The pH scale typically ranges from 0 to 14.
- Acidity:
- Substances with pH values less than 7 are considered acidic.
- The lower the pH value (closer to 0), the stronger the acid and the more acidic the solution. Extremely acidic substances have pH values below 1.
- Alkalinity (Basicity):
- Substances with pH values greater than 7 are considered alkaline (or basic).
- The higher the pH value (closer to 14), the stronger the base and the more alkaline the solution.
- Neutrality:
- A pH value of exactly 7 indicates a neutral solution (e.g., pure water at ).
pH and Hydrogen Ion Concentration
The pH value is an inverse logarithmic indication of the concentration of hydrogen ions (or hydronium ions, ) in a solution.
- Relationship:
- At pH 0, the hydronium ion concentration is ().
- At pH 14, the hydroxide ion concentration () is , which implies a very low concentration () in water.
- Formula: The pH is calculated using the formula: where is the molar concentration of hydrogen ions in moles per liter ().
- Inverse Relationship:
- A change of one pH unit represents a tenfold change in the concentration. For example, a solution with pH 3 has ten times the concentration of a solution with pH 4.
Worked Examples
Example 4.1: Finding pH from concentration
Find the pH of a solution of of .
- Given values:
- Concentration of
- Since is a strong acid, it dissociates completely, so .
- Therefore, .
- Apply the formula:
- Show calculation:
Example 4.2: Finding concentration from pH
If moist soil has a pH of 7.84, what is the concentration of the soil solution?
- Given values:
- Apply the formula (rearranged): From , we can derive .
- Show calculation:
- Hint: To calculate this, place -7.84 in your calculator and take the antilog (often inverse or ).
Possible Questions/Answers
Here are some practice questions based on the concepts discussed:
-
Q1: What is the pH of a solution of pure per of solution? A1:
- Molar mass of :
- Molarity of solution: Molarity =
- Calculate :
- Note: Phosphoric acid () is a weak acid. For a precise pH calculation, its acid dissociation constants ( values) would be needed. However, in many introductory contexts without explicit values, a simplified approach might be expected. If we assume it behaves as a strong monoprotic acid for estimation purposes (i.e., only the first proton dissociates completely), then .
- (This is an approximation. A more rigorous calculation for a weak polyprotic acid is more complex.)
-
Q2: Calculate the concentration of hydrogen ion () in a solution of sulphuric acid having pH of 1.5. A2:
- Given :
- Apply formula:
- Calculate :