2.4 Electrode, Electrode Potential and Electrochemical Series
This section explores the fundamental principles of electrochemistry, focusing on galvanic cells, electrode potentials, and the determination of redox reaction spontaneity.
2.4.1 The Galvanic Cell (Daniel Cell)
A galvanic cell, also known as a voltaic cell, is an electrochemical cell that converts chemical energy from spontaneous redox reactions into electrical energy.
Initial Observation
When a zinc (Zn) rod is placed directly into a copper(II) sulfate () solution, a spontaneous redox reaction occurs:
In this scenario, electrons flow directly from Zn to ions, and copper metal deposits on the zinc rod. No external current is generated.
Constructing a Galvanic Cell
To generate an electric current, the electron transfer must be directed through an external circuit.
Two separate containers are used: one with a Zn rod in zinc sulfate solution and another with a copper (Cu) rod in copper(II) sulfate solution. Initially, connecting the electrodes with a copper wire does not produce current because the circuits are not complete.
Salt Bridge
A salt bridge is a U-shaped tube filled with a solution of an inert electrolyte (e.g., , , ) that connects the two solutions.
The salt bridge allows the movement of ions between the two half-cells without mixing the solutions. It maintains electrical neutrality in both half-cells by allowing ions to flow, preventing charge buildup that would otherwise stop the reaction and current flow.
Types of Electrochemical Cells→
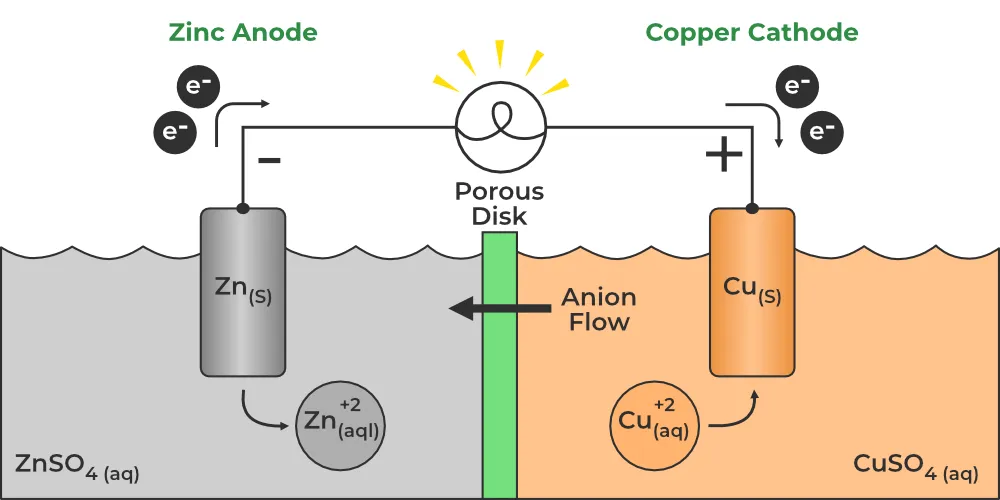
Half-Cells and Electrodes
The half-cell where oxidation occurs is called the anode half-cell, and the electrode is called the anode. The half-cell where reduction occurs is called the cathode half-cell, and the electrode is called the cathode.
Reactions in a Daniel Cell
Zinc has a greater tendency to lose electrons than copper. Thus, the Zn electrode becomes negatively charged relative to the Cu electrode. Electrons flow from the Zn electrode (anode) through the external circuit to the Cu electrode (cathode).
At the anode (oxidation half-reaction):
At the cathode (reduction half-reaction):
Overall cell reaction:
2.4.2 Cell Potential
Electromotive Force (emf)
The electromotive force is the "push" or "force" behind the electrons flowing from the anode to the cathode through the wire. It represents the potential of the cell to do work and is measured in volts (V).
Cell Potential ()
The emf produced by a galvanic cell depends on the difference in the electrode potentials of the two half-cells.
Under standard conditions ( concentration for solutions, pressure for gases, and ), the cell potential is denoted as . The electrode with a more negative standard reduction potential acts as the anode (oxidation). The electrode with a more positive standard reduction potential acts as the cathode (reduction).
Calculation of Standard Cell Potential
The standard cell potential is the algebraic difference between the standard reduction potentials of the cathode and anode:
For a spontaneous redox reaction, must have a positive value.
Measurement of Half-Cell Potentials
A single half-cell potential (electrode potential) cannot be measured directly because a half-cell reaction requires a simultaneous reaction in another half-cell. Relative half-cell potentials are determined by coupling the half-cell with a reference electrode, whose potential is arbitrarily set. The Standard Hydrogen Electrode (SHE) is the primary reference electrode.
2.4.3 Standard Hydrogen Electrode (SHE)
The SHE serves as the universally accepted reference electrode, with its potential arbitrarily assigned a value of at all temperatures.
Construction
A platinum (Pt) foil, coated with finely divided platinum (to increase surface area), is immersed in a hydrochloric acid () solution. Hydrogen gas () at pressure is bubbled around the platinum electrode at ().
Figure 2.2: Standard Hydrogen Electrode
Half-Reactions (Standard Potential = )
Reduction (acting as cathode):
Oxidation (acting as anode):
Flexibility
The SHE can function as either a cathode or an anode, depending on the other half-cell it is connected to.
Standard Conditions
The symbol indicates standard conditions: concentration for solutions, pressure for gases, and .
2.4.4 Method to Measure the Electrode Potentials
The standard electrode potential of a half-cell is determined by coupling it with the SHE.
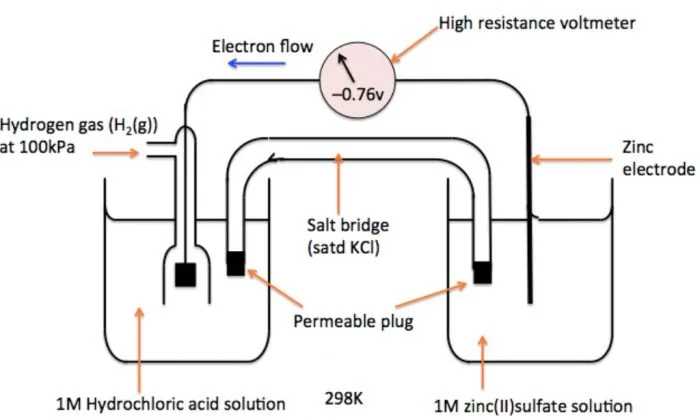
Determination of Standard Electrode Potential of Electrode
- Setup: A half-cell with a zinc rod in zinc sulfate solution is connected via a salt bridge to a SHE.
- Electron Flow: The voltmeter shows a reading of and indicates electron flow from the zinc electrode to the hydrogen electrode.
- Electrode Roles:
- Since electrons flow from the zinc electrode, zinc is undergoing oxidation and acts as the anode.
- The SHE undergoes reduction and acts as the cathode.
- Overall Cell Reaction:
- Calculations:
- Given .
- Using the formula :
- Since :
Determination of Standard Electrode Potential of Electrode
- Setup: A half-cell with a copper rod in copper sulfate solution is connected via a salt bridge to a SHE.
- Electron Flow: The voltmeter shows a reading of and indicates electron flow from the hydrogen electrode to the copper electrode.
- Electrode Roles:
- Since electrons flow to the copper electrode, copper ions are undergoing reduction, and copper acts as the cathode.
- The SHE undergoes oxidation and acts as the anode.
- Overall Cell Reaction:
- Calculations:
- Given .
- Using the formula :
- Since :
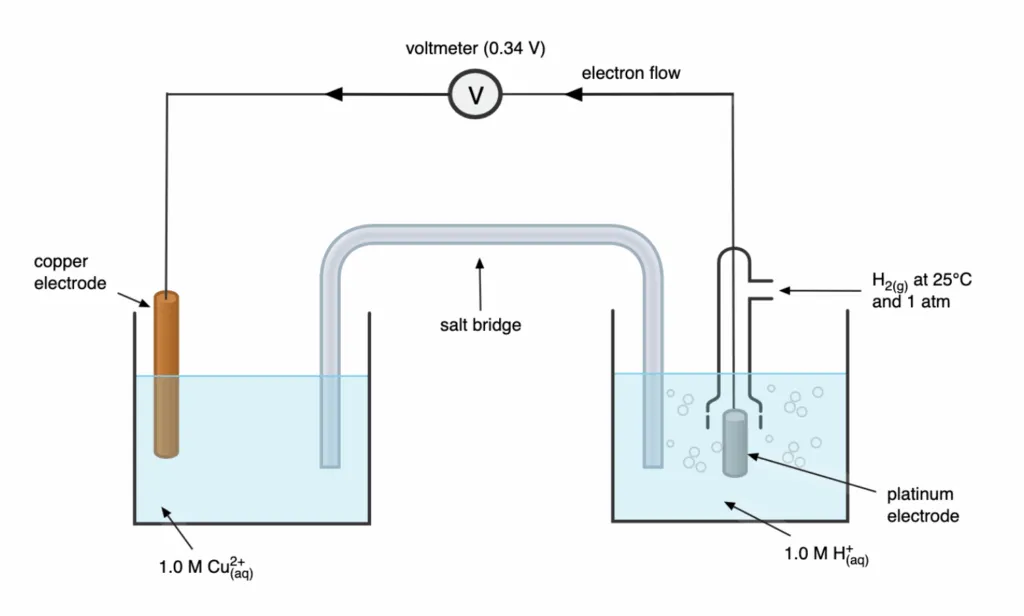
Table 2.1: Reduction Potentials of Some Elements, Ions and Compounds
| Reduction Half-reaction | (Volts) |
|---|---|
| -3.05 | |
| -2.92 | |
| -2.90 | |
| -2.76 | |
| -2.71 | |
| -2.38 | |
| -1.67 | |
| -1.03 | |
| -0.83 | |
| -0.76 | |
| -0.74 | |
| -0.44 | |
| -0.36 | |
| -0.25 | |
| -0.14 | |
| -0.13 | |
| -0.04 | |
| 0.00 | |
| +0.22 | |
| +0.27 | |
| +0.34 | |
| +0.52 | |
| +0.54 | |
| +0.77 | |
| +0.80 | |
| +1.09 | |
| +1.23 | |
| +1.28 | |
| +1.33 | |
| +1.36 | |
| +1.47 | |
| +1.49 | |
| +1.69 | |
| +1.70 | |
| +2.00 | |
| +2.87 |
2.4.4 Determination of Cell Potential (Revisited)
To determine the cell potential () for any galvanic cell:
- Identify Half-Reactions: Write down the two reduction half-reactions and their standard reduction potentials.
- Determine Anode/Cathode: The half-cell with the more positive reduction potential acts as the cathode. The more negative potential acts as the anode.
- Reverse Oxidation Half-Reaction: Reverse the anode half-reaction.
- Balance Electrons and Sum: Sum the two half-reactions. (Do not multiply values by coefficients).
- Calculate : Use .
2.4.5 Feasibility of a Chemical Reaction
The spontaneity (feasibility) of a redox reaction can be predicted from the sign of the standard cell potential ().
- If , the reaction is spontaneous.
- If , the reaction is non-spontaneous.
Worked Examples
Example 2.5: Calculate for the Zn-Cu Cell
1. Given values:
2. Determine roles:
- Cathode: Copper ()
- Anode: Zinc ()
3. Calculation:
Example 2.6: Calculate for the Ni-Mg Cell
1. Given values:
2. Determine roles:
- Cathode: Nickel ()
- Anode: Magnesium ()
3. Calculation:
Example 2.7: Is the Following Reaction Feasible?
1. Analyze reaction:
- Sn is oxidized (Anode), is reduced (Cathode).
2. Calculation:
3. Feasibility:
- Since is negative, the reaction is not feasible.
Possible Questions/Answers
CONCEPT ASSESSMENT EXERCISE 2.6
Q: Estimate for cell, write cell reactions, choose cathode and show the direction of electron flow.
A:
- Cathode: Fluorine ( is more positive).
- Anode: Copper.
- Reactions:
- Cathode:
- Anode:
- : .
- Electron Flow: From Cu (anode) to (cathode).
CONCEPT ASSESSMENT EXERCISE 2.7
Q1: Using emf data, argue on the following: (i) Can Mg displace Cu from a solution of Copper(II) sulphate? (ii) Can Iodine displace chlorine from an aqueous solution of Potassium chloride?
A1: (i) . Yes, Mg can displace Cu. (ii) For Iodine displacing Chlorine, . No, Iodine cannot displace Chlorine.