22.3 Energy Contents of Fuel
This section explores the energy stored in various materials, known as fuels. We will examine how this energy is quantified and compare different energy sources, including chemical fuels, nuclear energy, and solar energy.
Energy Density and Specific Energy
Many materials, such as food, coal, and petroleum, store energy and are collectively known as fuels. The energy content of these fuels is quantified using two key terms:
- Energy Density: The amount of energy stored in a fuel per unit volume.
- Unit: Joules per cubic meter ().
- Specific Energy: The amount of energy a fuel releases when it is burned per unit mass.
- Unit: Joules per kilogram () or watt-hours per kilogram (). For batteries, it is sometimes expressed as watts per hour per kg.
For example, the specific energy of coal is , while that of natural gas is significantly higher at .
Table 22.2: Specific Energy Density of Various Fuels
| Fuel Type | Reaction Type | Specific Energy Density (MJ/kg) | Typical Uses |
|---|---|---|---|
| Biodiesel | Chemical | 38 | Automotive engine |
| Coal | Chemical | 24 | Power plants, Electricity generation |
| Crude oil | Chemical | 44 | Refinery, Petroleum products |
| Diesel | Chemical | 45 | Diesel engines |
| Ethanol | Chemical | 26.8 | Gasoline mixture, Alcohol, Chemical products |
| Gasoline | Chemical | 46 | Gasoline engines |
| Natural gas | Chemical | 55 | Household heating, Electricity generation |
| Uranium-235 | Nuclear | 3,900,000 | Nuclear reactor electricity generation |
| Wood | Chemical | 16 | Space heating, Cooking |
Energy in Batteries
The efficiency of a battery is the ratio of the energy it can provide compared to the energy required to charge it. No battery is 100% efficient due to energy loss during storage, charging, and discharging. Lithium-ion batteries have a high efficiency of up to 99%, while lead-acid batteries are around 85% efficient.
Table 22.3: Energy Density of Batteries
| Storage Type | Specific Energy (MJ/kg) | Energy Density (MJ/L) |
|---|---|---|
| Battery, Lithium-ion | 0.46 - 0.72 | 0.83 - 3.6 |
| Battery, Lithium-Sulphur | 1.80 | 1.26 |
| Battery, Silver-oxide | 0.47 | 1.8 |
Nuclear Energy
Nuclear energy is the energy stored within the nucleus of an atom. As shown in Table 22.2, uranium can produce millions of times more energy than any chemical fuel. This energy is released through two primary processes: fission and fusion.
Nuclear Fission
Nuclear fission is the process where a heavy nucleus, like Uranium-235, splits into two or more smaller nuclei, releasing neutrons and a tremendous amount of energy.
Mechanism:
- A heavy nucleus (e.g., U-235 or Pu-239) absorbs a neutron.
- The absorption makes the nucleus excited and unstable.
- The unstable nucleus splits into two lighter nuclei and emits several free neutrons (typically 2-3).
- The total mass of the products is less than the mass of the reactants. This lost mass, known as the mass defect, is converted into energy according to Einstein's mass-energy equivalence equation:
Example Reaction:
The fission of Uranium-235:
Nuclear Fusion
Nuclear fusion is the process where two light atomic nuclei combine to form a heavier nucleus, releasing an enormous amount of energy.
Mechanism:
- Two light nuclei (e.g., isotopes of hydrogen like deuterium, ) are brought together under extremely high temperature and pressure.
- These conditions overcome the electrostatic repulsion between the positive nuclei, allowing them to fuse.
- The mass of the newly formed nucleus is less than the sum of the masses of the original nuclei. The mass difference is converted into energy.
Example Reactions:
Fusion of deuterium and tritium:
- Note: Fusion releases more energy than fission, produces non-toxic helium as a byproduct, and does not generate long-lived radioactive waste.
Importance of Nuclear Energy
- Provides a large amount of electricity from a small amount of fuel.
- Does not produce greenhouse gases () or other air pollutants during operation.
- Contributes to better air quality compared to fossil fuel power plants.
Solar Energy
The reliance on fossil fuels (coal, oil, natural gas) releases greenhouse gases that contribute to global warming and climate change. Transitioning to renewable energy sources like solar power is crucial for environmental sustainability.
Carbon Footprint
A Carbon Footprint is the total amount of greenhouse gases (especially ) emitted directly or indirectly by a human activity, organization, or product. It is typically measured in tons of equivalent per year. Reducing our carbon footprint is essential to combat climate change.
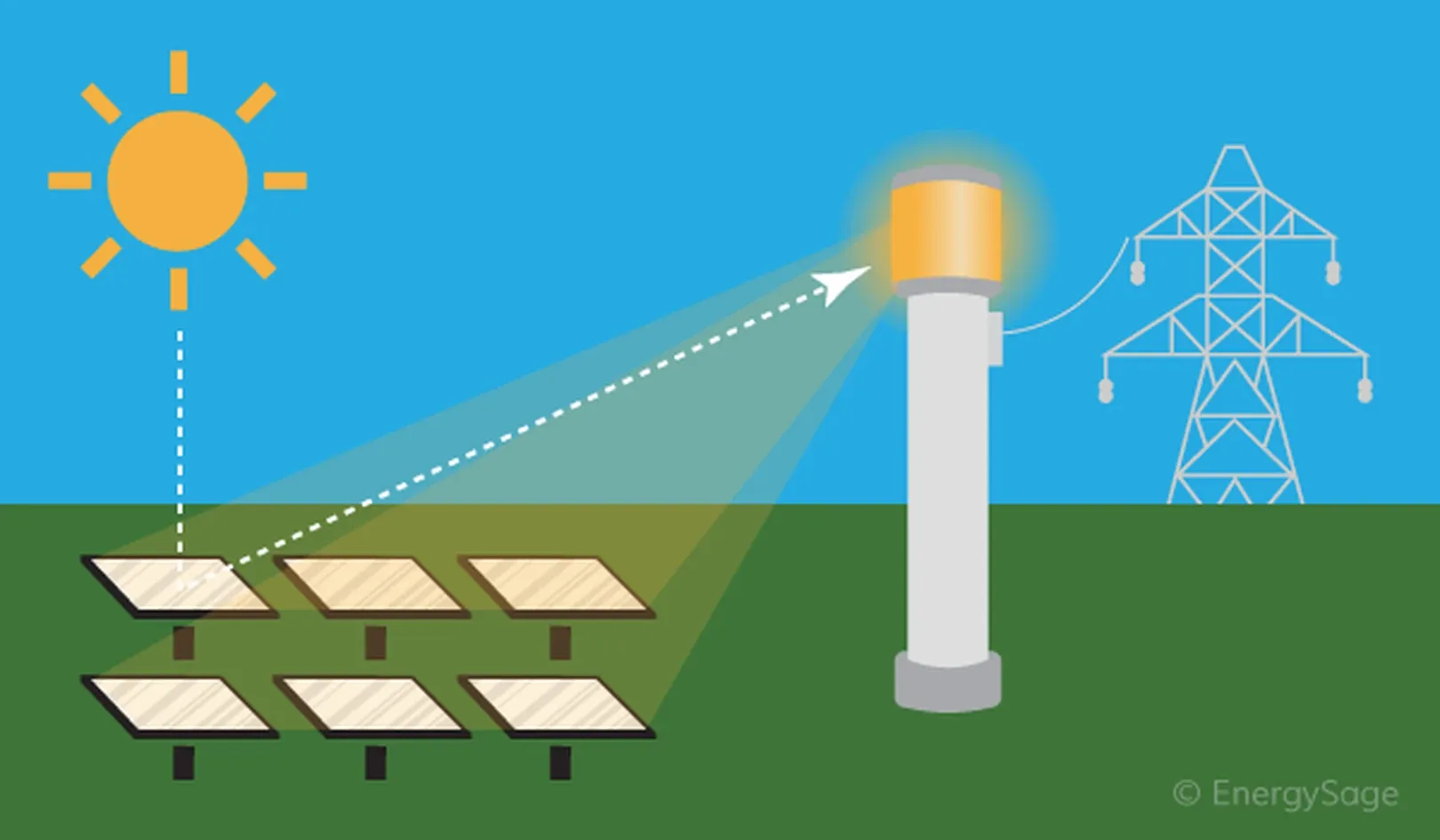
Mechanism of Solar Energy Conversion
Solar panels capture sunlight and convert it into usable energy (heat or electricity) through two main technologies:
- Solar Thermal Power (CSP) Systems:
- These systems use mirrors or lenses to concentrate sunlight onto a receiver.
- The receiver contains a fluid (liquid or oil) that gets heated.
- This heat can be used directly for heating or to generate steam to drive a turbine and produce electricity.
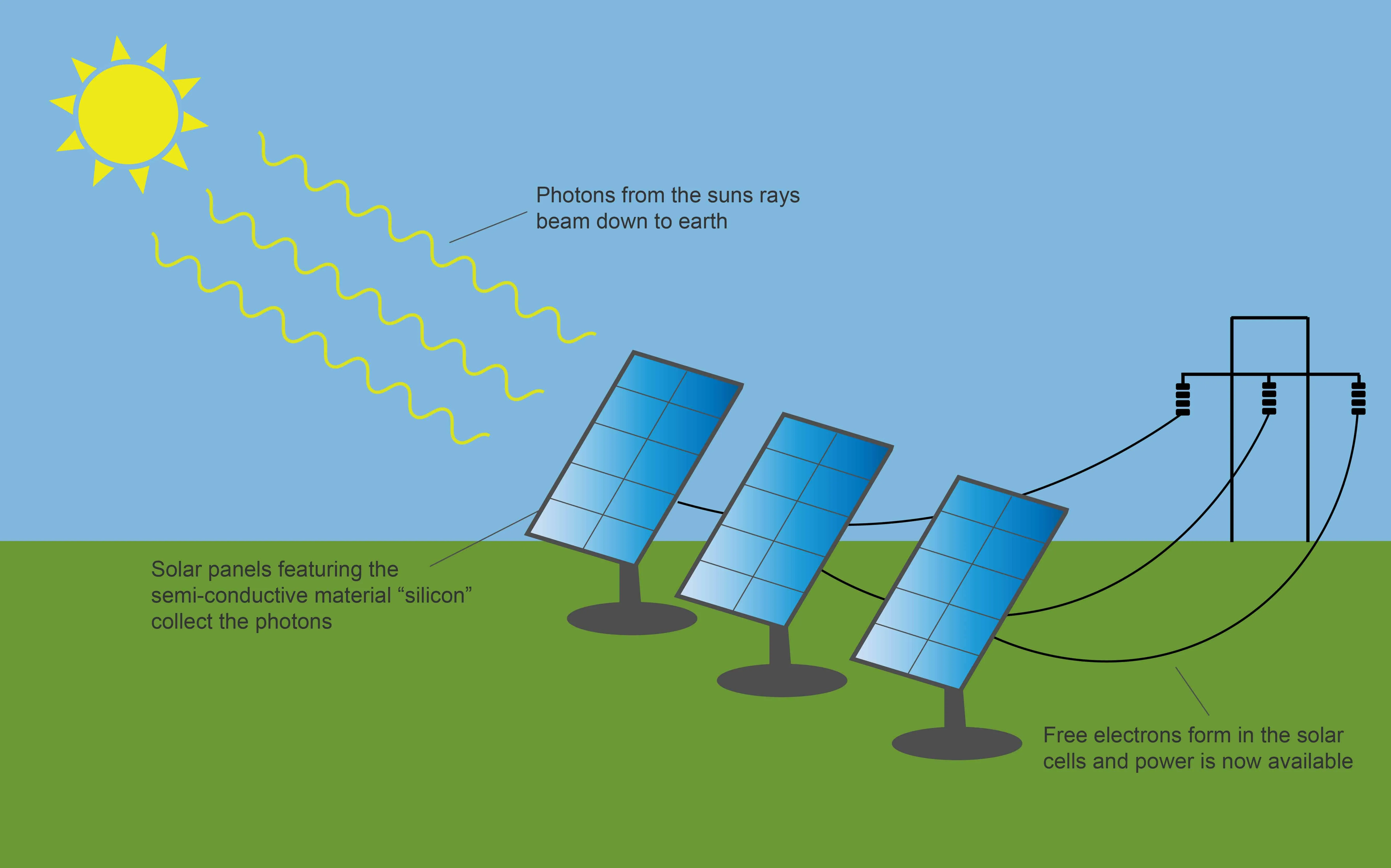
- Photovoltaic (PV) Cells:
- PV cells directly convert sunlight into electricity.
- When sunlight (photons) strikes the semiconductor material (e.g., silicon) in a PV cell, it excites electrons, freeing them from their atoms.
- This electron flow creates an electric current, specifically a direct current (DC).
- An inverter converts the DC electricity into alternating current (AC), which is used to power homes and businesses.
Advantages of Photovoltaic Cells
- Renewable Energy Source: Solar energy is abundant and inexhaustible.
- Low Environmental Impact: Produces no air or water pollution during operation.
- Reduced Greenhouse Gas Emissions: A clean energy source that helps reduce carbon footprints.
- Reduced Dependency on Fossil Fuels: Provides energy independence.
- Long Lifespan and Low Maintenance: Solar panels typically last 25 years or more with minimal upkeep.
Comparison of Energy Sources
Table 22.4: Advantages and Disadvantages of Different Energy Sources
| Energy Resource | Advantages | Disadvantages |
|---|---|---|
| Fossil fuels | Reliable and readily available. | Creates pollution (greenhouse gases, acid rain) and is a finite resource. |
| Nuclear power | Reliable, produces vast energy from little fuel, low emissions. | Fuel is finite, produces dangerous radioactive waste that is difficult to dispose of. |
| Wind turbines | Clean energy, cheap to operate. | Expensive initial setup, dependent on wind, can be noisy and visually obtrusive. |
| Hydroelectric power | Clean energy, cheap to operate. | Expensive initial setup, can be affected by drought, impacts river ecosystems. |
| Solar cells | Clean energy, cheap to operate. | Dependent on sunlight (not always sunny), initial setup cost can be high. |
A balanced and sustainable energy future will likely involve a combination of these sources to meet global energy needs while minimizing environmental impact.