22.1 Origin of Petroleum and Natural Gas
It is widely accepted that petroleum, also known as crude oil, and natural gas originated from organisms that existed hundreds of millions of years ago.
- Formation Process:
- Marine plants and animals died, and their remains accumulated.
- These remains were subsequently buried under layers of sand, rock, and mud.
- Over extensive periods, and in the absence of air, the combined effects of heat and pressure from the overlying sediments, along with the activity of bacteria, transformed the organic material into a dark brown, viscous liquid known as petroleum.
- Natural gas consists of gas products that accumulate above the oil deposits.
22.1.1 Mining of Petroleum
The extraction of crude oil from the Earth's crust involves a multi-step drilling process:
- Drilling: Holes are drilled into the earth where petroleum deposits are found.
- Natural Gas Release: When drilling penetrates the rock layers, natural gas, which is often under high pressure, typically emerges first.
- Oil Extraction: Once the natural gas pressure subsides, the crude oil is then extracted using powerful pumps.
22.1.2 Refining of Petroleum
The process of converting crude oil into useful products is called refining. The beneficial products obtained are referred to as fractions, each consisting of a collection of hydrocarbons that boil within a specific temperature range.
Fractional Distillation of Petroleum
Petroleum is refined through a process called fractional distillation, which takes place in a tall fractionating tower (See Figure 22.1 below).
- Process Steps:
- Heating: Crude oil is initially heated in a furnace to approximately .
- Vaporization: The heated crude oil, now largely vaporized, is passed into the bottom of the fractional distillation column.
- Ascension and Condensation: The hot vapors rise through the column. As they ascend, they cool down. Compounds with different boiling points condense at different levels within the column:
- Compounds with very high boiling points condense first and collect near the bottom of the column.
- Compounds with the lowest boiling points continue to rise and condense last, collecting near the top of the column.
- Compounds that do not boil remain as a residue at the very bottom.
- Separation: This gradual condensation separates the crude oil into various fractions, each characterized by its own specific boiling range and chemical composition.
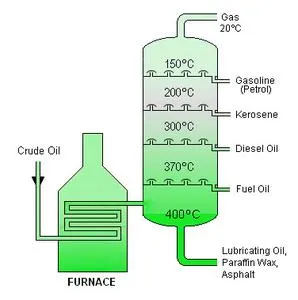
Petroleum Fractions and Their Uses
The table below summarizes the physical properties and important uses of the main petroleum fractions obtained from fractional distillation.
Table 22.1: Fractions of petroleum and their uses
| Fraction | Number of carbon atoms per molecule | Boiling point | Important uses |
|---|---|---|---|
| Liquefied petroleum gas (LPG) | 1–4 | Below 20 | Cylinder gas for cooking |
| Petrol | 5–10 | 35–70 | Fuel for motor cars and vehicles |
| Naphtha | 8–12 | 70–120 | Chemical feedstock for making drugs, plastics, and other chemicals |
| Kerosene | 10–16 | 170–250 | Fuel for jet planes, fuel for heating, lighting, and cooking |
| Diesel | 14–20 | 270–340 | Fuel for buses, trucks, and trains |
| Lubricating oil | 20–50 | 350–500 | Lubricants for machines and engines, waxes, and polishes |
| Fuel oil | 50–70 | 500–600 | Fuel for power stations, factories, and ships |
| Bitumen | More than 70 | More than 500 | Paving roads and making roofing materials |