18.1 Classification of Monohydric Alcohols
Monohydric alcohols contain only one hydroxyl (-OH) group per molecule. They are classified into three main types based on the number of alkyl groups attached to the carbinol carbon — the carbon atom directly bonded to the -OH group.
Classification of Alcohols
Monohydric alcohols are categorized into:
- (a) Primary (1°) alcohols
- (b) Secondary (2°) alcohols
- (c) Tertiary (3°) alcohols
The general formula for saturated monohydric alcohols is (or ).
(a) Primary Alcohols
In primary (1°) alcohols, the carbinol carbon is bonded to only one other alkyl group (or no alkyl groups, as in methanol).
- General Formula:
- Examples:
- Methanol:
- Ethanol:
- 1-Propanol:
Ethyl alcohol (Primary alcohol)
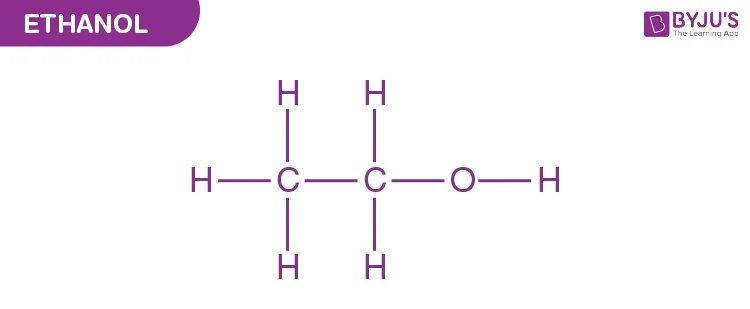
(b) Secondary Alcohols
In secondary (2°) alcohols, the carbinol carbon is bonded to two alkyl groups (same or different).
- General Formula:
- Example:
- 2-Propanol (Isopropyl alcohol):
Isopropyl alcohol (Secondary alcohol)
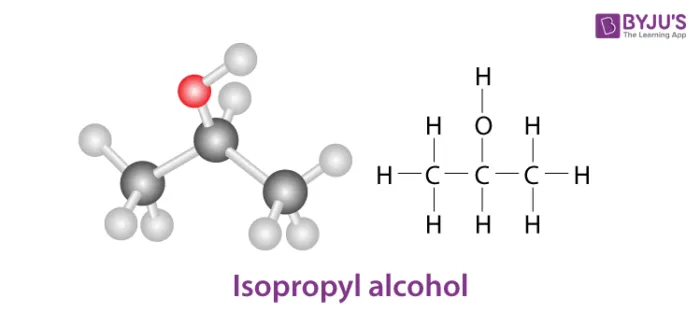
(c) Tertiary Alcohols
In tertiary (3°) alcohols, the carbinol carbon is bonded to three alkyl groups (same or different).
- General Formula:
- Example:
- 2-Methyl-2-propanol (Tert-butyl alcohol):
Tert-butyl alcohol (Tertiary alcohol)
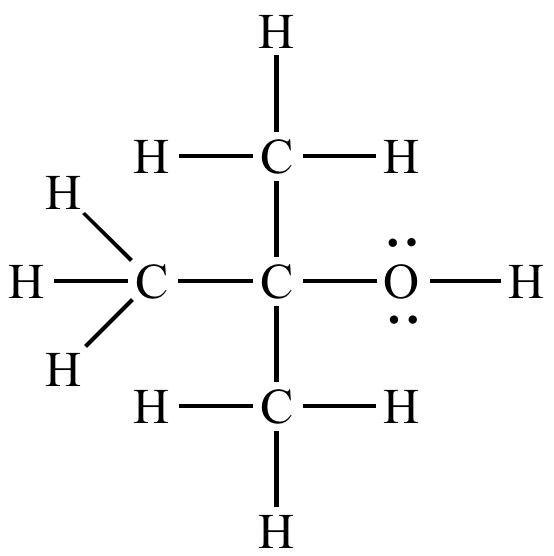
Summary Table
| Alcohol Class | General Structure | Carbinol Carbon bonded to | Example |
|---|---|---|---|
| Primary (1°) | One alkyl group | Ethanol () | |
| Secondary (2°) | Two alkyl groups | 2-Propanol () | |
| Tertiary (3°) | Three alkyl groups | 2-Methyl-2-propanol () |
Note: This classification is important because the reactivity of alcohols — including oxidation behaviour and reaction with Lucas reagent — depends on whether they are primary, secondary, or tertiary.