16.7 Conjugation in Alkenes
Definition of Conjugation
The term conjugation originates from the Latin for "to join together." In chemistry, it describes a system where unhybridized p-orbitals on adjacent atoms in a molecule overlap, creating a continuous "bridge" for pi () electrons.
Conjugation is a key feature in alkenes that possess alternating double and single bonds. In these molecules:
- The carbon atoms involved in the alternating system are hybridized.
- Each of these carbon atoms has one unhybridized p-orbital that is perpendicular to the plane of the sigma () bonds.
Delocalization of Pi () Electrons
The side-by-side overlap of these unhybridized p-orbitals creates a larger, continuous molecular orbital that extends across the entire conjugated system.
- The double bonds in such molecules are described as delocalized.
- This means their electrons are not confined between two specific carbon atoms but can move freely along the entire bridge of overlapping p-orbitals.
- Delocalization generally leads to increased molecular stability (resonance stabilization energy).
Bond Length Evidence for Conjugation
Conjugation has a measurable effect on bond lengths. In 1,3-butadiene ():
| Bond | Length |
|---|---|
| C=C double bond | |
| C–C single bond (C2–C3) | |
| Typical alkane C–C bond |
The central C2–C3 single bond is shorter than a typical alkane C–C bond because:
- Both C2 and C3 are hybridized (stronger overlap than –).
- Partial double-bond character arises from electron delocalization across all four carbons.
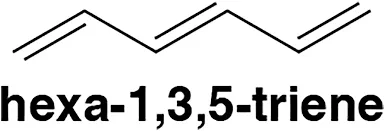
Example: Hexa-1,3,5-triene
Hexa-1,3,5-triene is a classic example of a conjugated system.
- Structure:
- The molecule features an alternating pattern of three double bonds and two single bonds.
Concept Assessment Exercise 16.5
Question: Explain which one of the following compounds shows conjugation. i. ethene ii. but-1-ene iii. penta-1,2-diene iv. buta-1,2-diene v. penta-1,3-diene
Answer and Explanation:
To determine if a compound shows conjugation, we look for an alternating pattern of double and single bonds.
-
i. Ethene ()
- This molecule contains only one double bond. There is no alternating system.
- Conclusion: Not conjugated.
-
ii. But-1-ene ()
- This molecule has one double bond and two single bonds in the chain, but they are not in an alternating sequence. The double bond is isolated.
- Conclusion: Not conjugated.
-
iii. Penta-1,2-diene ()
- The double bonds are adjacent (cumulative), not separated by a single bond. This type of compound is called an allene.
- Conclusion: Not conjugated.
-
iv. Buta-1,2-diene ()
- Like penta-1,2-diene, this molecule has cumulative double bonds. It is also an allene.
- Conclusion: Not conjugated.
-
v. Penta-1,3-diene ()
- This molecule's structure shows a double bond, followed by a single bond, followed by another double bond. This is the required alternating pattern.
- Conclusion: Shows conjugation.
Summary
- Conjugation is a property of molecules that have an alternating sequence of single and double bonds.
- It arises from the overlap of unhybridized p-orbitals on adjacent hybridized carbon atoms.
- This overlap forms a continuous molecular orbital system, allowing electrons to be delocalized across multiple atoms.
- Delocalization leads to increased molecular stability and measurably shorter central C–C single bonds (e.g., in 1,3-butadiene vs in alkanes).
- A molecule like penta-1,3-diene () is conjugated, while molecules with isolated () or cumulative () double bonds are not.