13.1 AIR POLLUTION
The environment consists of various interconnected components that shape the natural world: the atmosphere (air), hydrosphere (water), lithosphere (land), and biosphere (living organisms).
- Atmosphere: The gaseous envelope surrounding the Earth, primarily composed of nitrogen (, ~78%) and oxygen (, ~21%), with trace gases like carbon dioxide (), water vapor, and noble gases. It regulates temperature, weather, and protects life from solar radiation.
- Hydrosphere: Encompasses all of Earth's water, including oceans, rivers, lakes, glaciers, and groundwater. It is vital for life and influences climate by distributing heat.
- Lithosphere: The solid, outermost layer of the Earth, including the crust, continents, and ocean beds. It is composed of rocks, minerals, and soils and is the site of geological processes.
- Biosphere: Includes all living organisms and their interactions within their ecosystems.

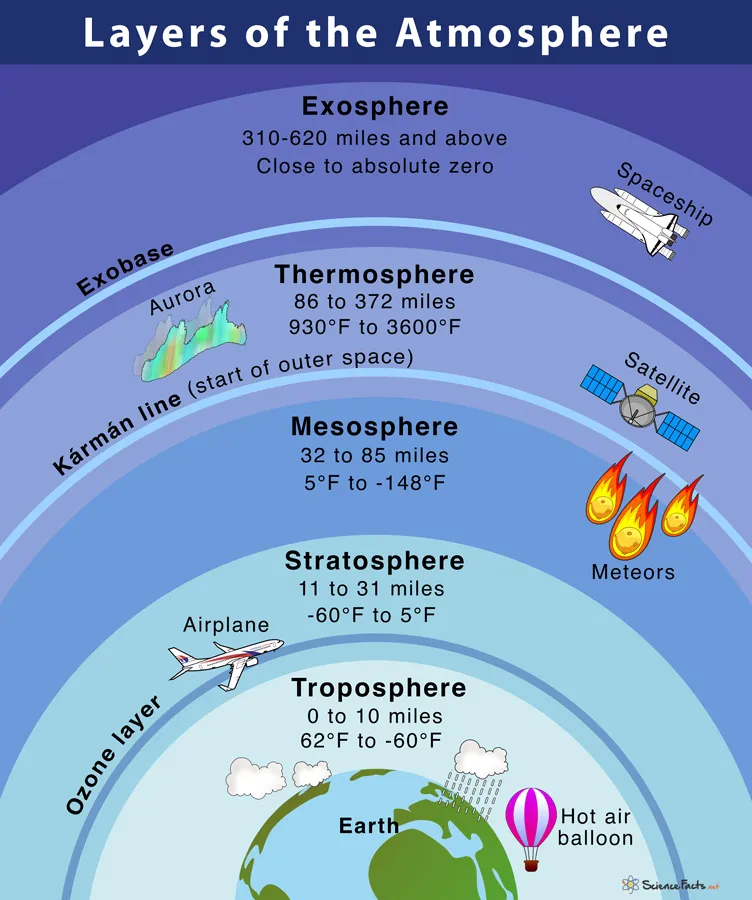
13.1.1 Atmosphere and its Layers
The Earth's atmosphere is structured into four main layers based on temperature variations.
-
Troposphere:
- The lowest layer, extending from the Earth's surface up to approximately 12 km.
- Contains about 78% nitrogen and 21% oxygen.
- It is where weather phenomena, clouds, and water vapor are concentrated.
- Temperature decreases with increasing altitude.
-
Stratosphere:
- Located above the troposphere.
- Contains the ozone layer (), which absorbs harmful ultraviolet (UV) radiation from the sun.
- Temperature increases with altitude due to the absorption of UV radiation by ozone.
-
Mesosphere:
- The layer above the stratosphere, characterized by extremely low temperatures.
- This is where most meteors burn up upon entering the atmosphere.
- Auroras can occur in this layer.
-
Thermosphere:
- The outermost layer, characterized by very high temperatures due to the absorption of intense solar radiation.
- It is divided into two sub-layers:
- Ionosphere: Contains electrically charged particles (ions) that are crucial for radio communication.
- Exosphere: The final frontier that gradually merges into space. It contains very thin gases like hydrogen and helium, and particles can escape Earth's gravity.
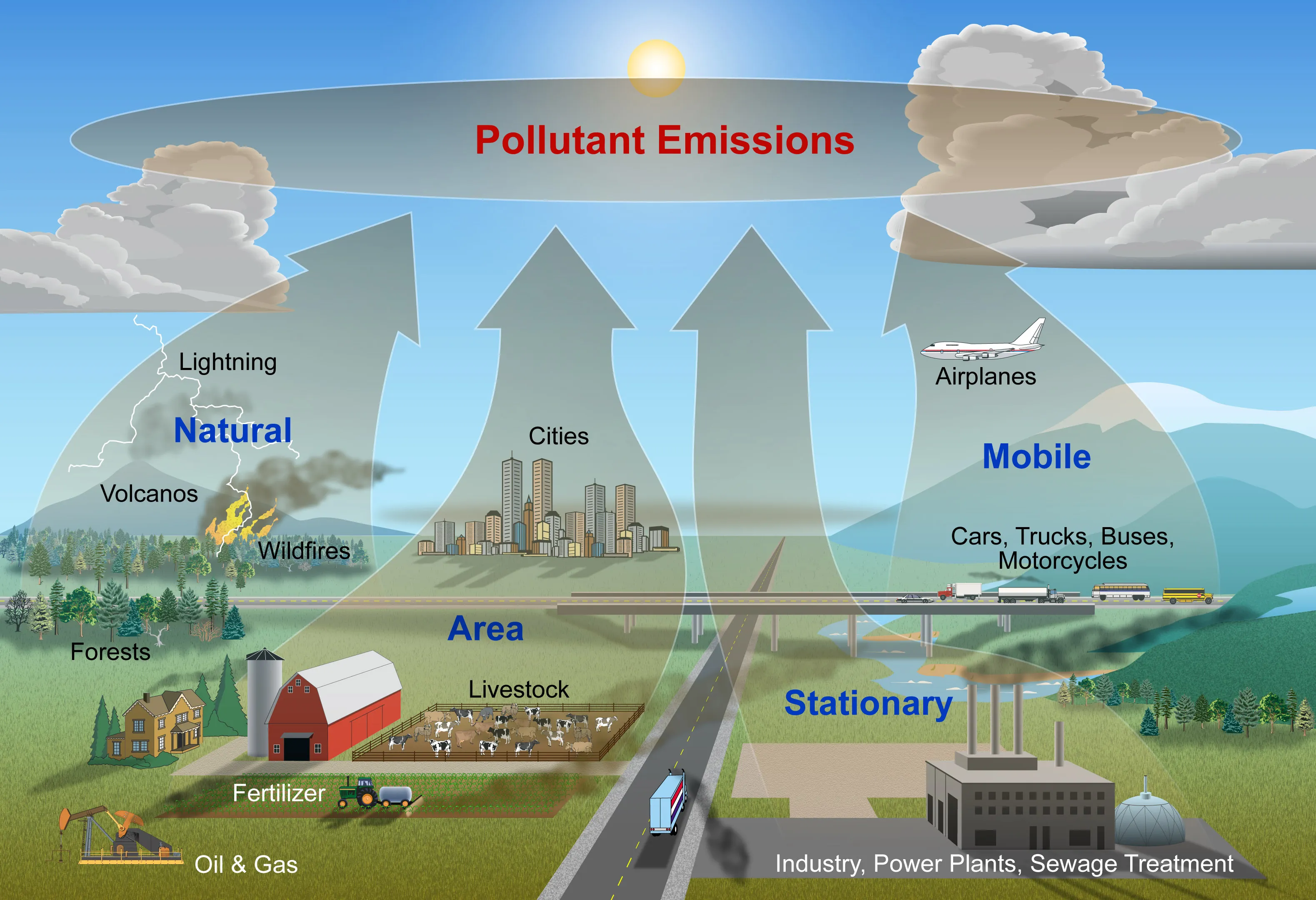
13.1.2 Air Pollutants
Air pollutants are substances in the air that can be harmful to humans and the environment. They are categorized as primary pollutants (emitted directly from a source) and secondary pollutants (formed from chemical reactions in the atmosphere).
Common Air Pollutants
- Particulate Matter (PM): Tiny solid or liquid particles from combustion, construction, and natural sources. They can cause respiratory and cardiovascular problems.
- Nitrogen Oxides (): Gases produced from burning fossil fuels in vehicles and industries. They contribute to smog and acid rain.
- Sulphur Dioxide (): Primarily emitted from burning coal and oil. It causes respiratory issues and is a major precursor to acid rain.
- Volatile Organic Compounds (VOCs): Released from vehicle emissions, industrial processes, and household products. They are key components in the formation of ground-level ozone.
- Carbon Monoxide (): A toxic gas from incomplete combustion. It reduces the oxygen-carrying capacity of blood.
- Ground-Level Ozone (): A secondary pollutant formed when and VOCs react in sunlight. It is a major component of smog and harms the respiratory system.
- Heavy Metals: Metals like lead (), mercury (), and cadmium () released from industrial processes. They are toxic and can accumulate in the food chain.
- Ammonia (): Emitted from agricultural activities. It contributes to particulate matter formation.
- Polycyclic Aromatic Hydrocarbons (PAHs): Organic compounds with multiple aromatic rings, formed during incomplete combustion of materials like coal, oil, and wood. Many are carcinogenic.
- Chlorofluorocarbons (CFCs): Synthetic compounds once used in refrigeration and aerosols. They are potent greenhouse gases and are responsible for the depletion of the stratospheric ozone layer.
Does the Hole in the Ozone Layer Still Exist?
The stratospheric ozone layer protects us from dangerous UV radiation. During the 1980s and 1990s, the extensive use of CFCs caused a significant thinning, often called a "hole," in this layer. International agreements like the Montreal Protocol have phased out CFCs, and the ozone layer is slowly recovering, but it is a long-term process. For more on the chemistry of halogens used in CFCs, see 12.1 Halogens→(/chemistry-11/12-halogens/12-1-halogens).
Summary of Air Pollutants
| Pollutant Category | Examples | Physical State | Major Sources | Health/Environmental Effects |
|---|---|---|---|---|
| Particulate Matter (PM) | Dust, fly ash, soot | Solid/Liquid | Combustion, construction, mining | Respiratory and cardiovascular disease |
| Nitrogen Oxides | , | Gas | Vehicle engines, power plants | Smog, acid rain, respiratory irritation |
| Sulphur Dioxide | Gas | Coal/oil combustion, ore smelting | Acid rain, respiratory problems | |
| Carbon Monoxide | Gas | Incomplete combustion | Reduces blood oxygen capacity; toxic | |
| Volatile Organic Compounds | Benzene, toluene | Gas/Vapour | Vehicles, solvents, paints | Ground-level ozone formation, carcinogenic |
| Ground-Level Ozone | Gas | Secondary (from + VOCs + sunlight) | Lung damage, crop damage | |
| Heavy Metals | , , | Solid/Aerosol | Industrial processes, incineration | Neurotoxic, bioaccumulation in food chain |
| Chlorofluorocarbons | CFCs | Gas | Refrigerants, aerosols (historical) | Ozone layer depletion, greenhouse effect |
| Ammonia | Gas | Agriculture, livestock | Particulate matter formation, ecosystem damage | |
| PAHs | Benzo[a]pyrene | Solid/Aerosol | Incomplete combustion of organic matter | Carcinogenic |