8.2 Reversible Reactions and Dynamic Equilibrium
This section explores the nature of chemical reactions that can proceed in both forward and reverse directions, leading to a state of dynamic equilibrium where the concentrations of reactants and products remain constant.
Reversible Reactions
A reversible reaction is a chemical reaction where the products, once formed, can react with each other to re-form the original reactants. These reactions do not proceed to completion, even with stoichiometric amounts of reactants, because they occur simultaneously in both the forward and backward directions.
- The direction from reactants to products is the forward reaction.
- The direction from products back to reactants is the reverse reaction.
- A double arrow () is used in chemical equations to indicate that a reaction is reversible.
- Reversible reactions must occur in a closed system so that neither reactants nor products can escape, allowing both directions to proceed simultaneously.
Examples of Reversible Reactions:
Achieving Dynamic Equilibrium
Consider the reaction between steam () and carbon monoxide () to understand how equilibrium is established.
1. The Forward Reaction
- When steam and carbon monoxide are first mixed, their concentrations are at a maximum.
- The initial rate of the forward reaction is at its highest because the frequency of collisions between reactant molecules is greatest.
- As the reaction proceeds, the concentrations of and decrease, causing the rate of the forward reaction to slow down.
2. The Reverse Reaction
- As the products, hydrogen () and carbon dioxide (), accumulate, they begin to collide and react to form the original reactants.
- This reverse reaction starts at a rate of zero and gradually increases its speed as the product concentrations rise.
3. The State of Equilibrium
- Eventually, a point is reached where the rate of the forward reaction becomes equal to the rate of the reverse reaction.
- At this stage, the reaction is said to be in chemical equilibrium.
- The concentrations of all reactants and products become constant and no further macroscopic changes are observed.
Chemical Equilibrium: The state of a reversible reaction at which the composition of the reaction mixture does not change is called the state of chemical equilibrium.
The graph below illustrates how the concentrations of reactants and products change over time until equilibrium is reached.
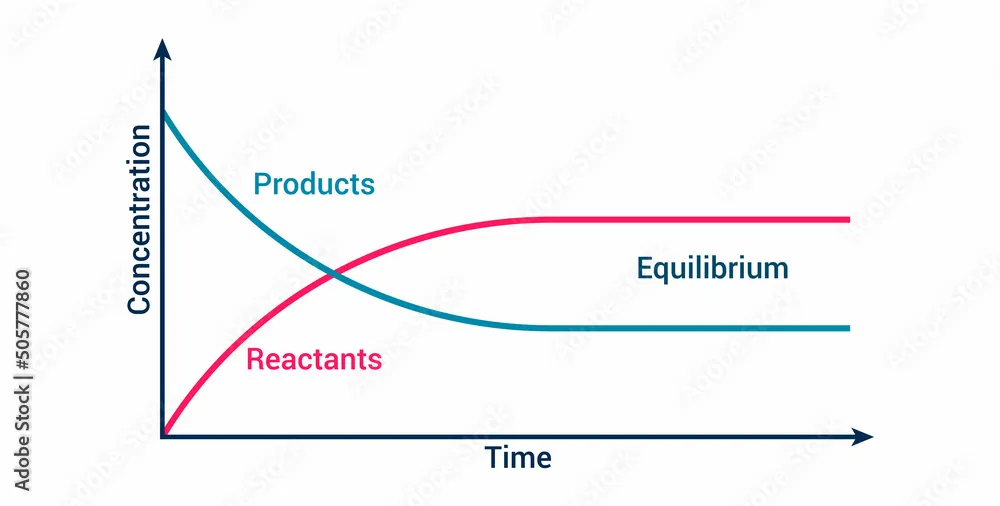
The Nature of Dynamic Equilibrium
Although the overall concentrations appear constant at equilibrium, the reaction has not stopped. At the microscopic level, there is intense activity:
- Individual reactant molecules continue to combine to form products.
- Individual product molecules continue to combine to re-form reactants.
This state is called dynamic equilibrium because the system is dynamic (molecules are constantly reacting), but it is balanced (the forward and reverse reaction rates are equal). There is no net change in the overall composition of the mixture.
Key features of dynamic equilibrium:
| Feature | Description |
|---|---|
| Macroscopic | Concentrations, pressure, colour — all constant |
| Microscopic | Forward and reverse reactions continue at equal rates |
| System requirement | Must be a closed system |
| Temperature | Must remain constant |
See also for the distinction between observable changes and molecular-level activity.
<PracticeQuestions questionsString="[{"question":"What is a reversible reaction?","answer":"A chemical reaction where the products can react together to reform the original reactants, denoted by a double arrow.","flash