7.5 Activation Energy
This section explores the energy requirements for chemical reactions, focusing on collision theory and the activated complex.
Collision Theory
For a chemical reaction to occur, reactant particles must collide. However, not all collisions result in a reaction. Collision theory explains the factors that determine whether a collision will be successful.
A collision is considered effective (leads to a reaction) only if two conditions are met:
- Sufficient Energy: The colliding particles must possess a minimum amount of energy to overcome the repulsive forces between their electron clouds.
- Proper Orientation: The particles must be oriented correctly at the moment of collision so that the atoms required to form new bonds come into direct contact.
Collisions that do not meet these two criteria are ineffective, and the particles simply bounce off each other chemically unchanged.
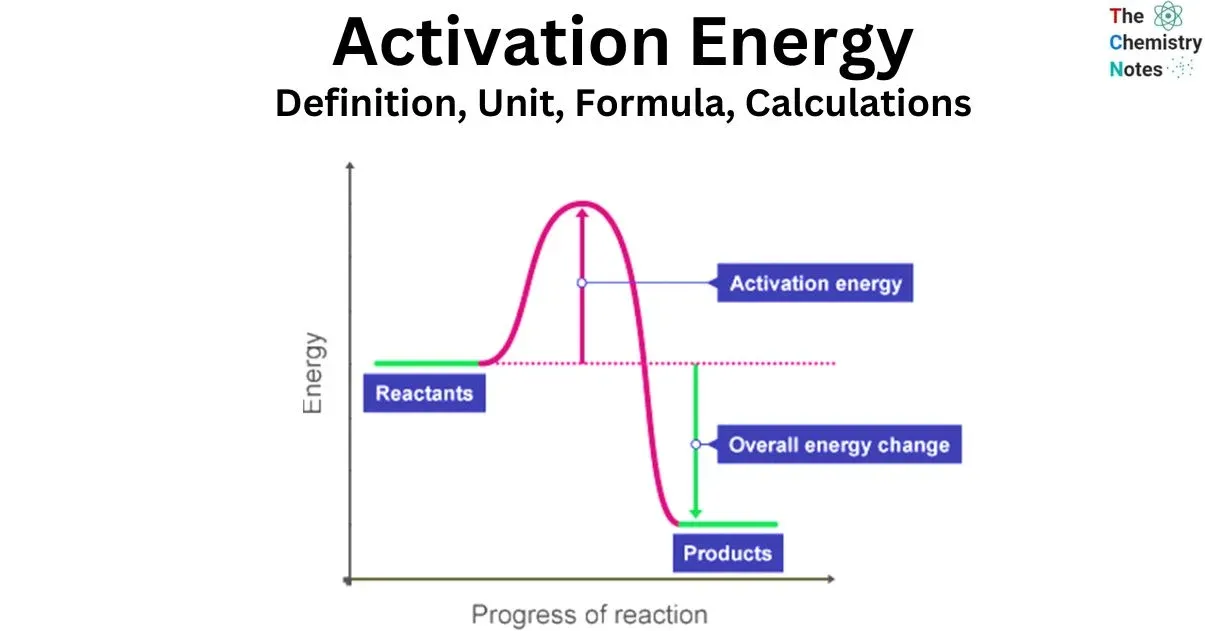
Activation Energy ()
Activation Energy () is defined as the minimum amount of energy, in addition to the average kinetic energy, that colliding particles must possess for an effective collision to occur.
- It acts as an energy barrier that must be overcome for reactants to be converted into products.
- If the energy of the colliding particles is less than the activation energy, no reaction will take place.
Relationship with Reaction Rate:
- High Activation Energy: A higher energy barrier means that only a small fraction of molecules will have sufficient energy to react upon collision. This results in a slower reaction rate.
- Low Activation Energy: A lower energy barrier allows a larger fraction of molecules to undergo effective collisions, leading to a faster reaction rate.
Do You Know? Gas explosions in homes can be caused by switching on a light. If gas has been leaking, then a tiny spark from turning on a light can provide the activation energy to start the explosive reaction between the methane and the oxygen.
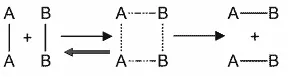
The Activated Complex (Transition State)
When reactants with sufficient energy and proper orientation collide, they form a temporary, unstable, high-energy species called the activated complex or transition state.
- Formation: During an effective collision, the kinetic energy of the reactants decreases as they slow down, and this energy is converted into potential energy, leading to the formation of the activated complex.
- Characteristics: The activated complex is highly unstable and short-lived.
- Breakdown: It quickly breaks down to form the more stable products of the reaction.
For example, in the reaction between and :
Here, represents the activated complex.
Potential Energy Diagrams
A potential energy diagram illustrates the energy changes that occur during a chemical reaction. The activation energy () is represented as an energy hill that reactants must "climb" to become products.
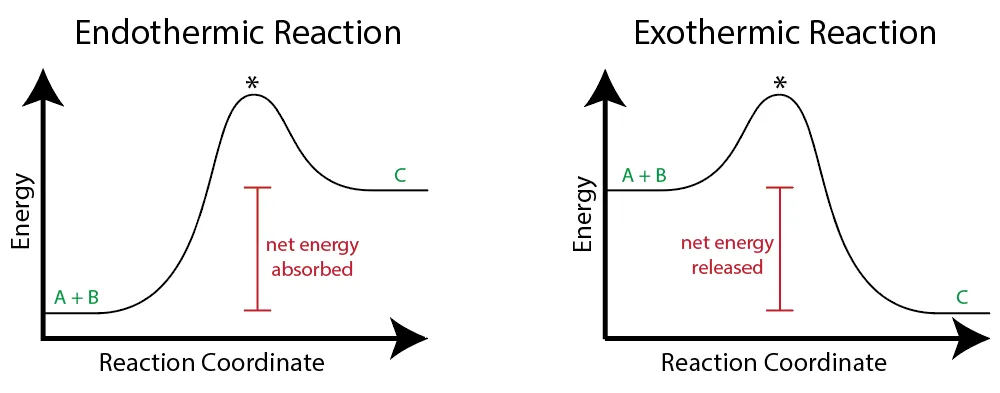
1. Exothermic Reactions
- The products are at a lower potential energy level than the reactants.
- Energy is released into the surroundings.
- The enthalpy change () is negative.
- The activation energy for the reverse reaction is higher than for the forward reaction ().
2. Endothermic Reactions
- The products are at a higher potential energy level than the reactants.
- Energy is absorbed from the surroundings.
- The enthalpy change () is positive.
- These reactions often require a continuous supply of energy to proceed.
In both cases, the activation energy () is the energy difference between the reactants and the peak of the energy barrier (the activated complex). Adding a catalyst provides an alternative pathway with a lower activation energy.