7.3 Effect of Temperature on the Rate of Reactions
The rate of a chemical reaction is highly dependent on temperature. Generally, an increase in temperature leads to an increase in the reaction rate.
Collision Theory and Temperature
According to collision theory, a reaction occurs when reactant molecules collide with sufficient energy and in the correct orientation. The effect of temperature can be explained by this theory:
- Increased Kinetic Energy: An increase in temperature raises the average kinetic energy of the molecules.
- Increased Molecular Speed: As molecules gain kinetic energy, their average speed increases.
- Increased Collision Frequency: Faster-moving molecules collide more frequently.
However, not all collisions result in a reaction. For a collision to be effective, two conditions must be met:
- The colliding molecules must possess a minimum amount of energy, known as the activation energy ().
- The molecules must be in the correct spatial orientation at the moment of impact.
Key insight: Collision frequency increases by only about – for a rise in temperature. The dominant reason for the sharp increase in reaction rate is the exponential increase in the fraction of molecules possessing energy .
The Maxwell-Boltzmann Distribution
At any given temperature, the reactant molecules do not all have the same kinetic energy. The Maxwell-Boltzmann distribution curve illustrates how kinetic energy is distributed among a population of molecules at a constant temperature.
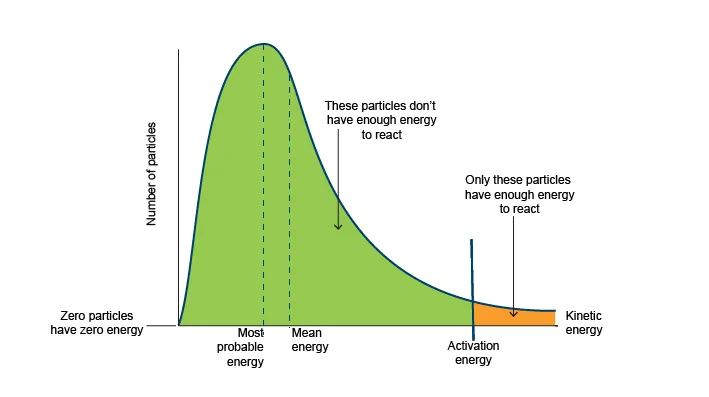
- The peak of the curve represents the most probable kinetic energy.
- The activation energy () is a threshold on this curve. Only molecules with kinetic energy equal to or greater than can react upon collision.
- The shaded area under the curve to the right of represents the fraction of molecules with sufficient energy to react.
Effect of Increasing Temperature
When the temperature is increased (e.g., from to , where ):
- The distribution curve flattens and shifts to the right.
- The fraction of molecules possessing the required activation energy () increases significantly.
- This leads to a higher number of effective collisions per unit time, thereby increasing the reaction rate.
Temperature Coefficient ()
A general rule of thumb is that for many reactions, the rate doubles or triples for every 10 K (or 10°C) increase in temperature. This is quantified by the Temperature Coefficient ():
For most reactions, –.
The Arrhenius Equation
In 1889, Svante Arrhenius quantitatively described the relationship between temperature, activation energy, and the rate constant () with the Arrhenius equation:
Where:
| Symbol | Meaning |
|---|---|
| Rate constant | |
| Pre-exponential factor (related to collision frequency and orientation probability) | |
| Activation energy (J/mol) | |
| Universal gas constant () | |
| Absolute temperature (Kelvin) |
This equation shows that increases exponentially as increases. Since reaction rate is directly proportional to , the reaction rate also increases with temperature.
Linearised Form
Taking the natural logarithm of the Arrhenius equation:
A plot of vs gives a straight line with:
- Slope
- Intercept
This allows experimental determination of from kinetic data.