7.1 Rates of Reactions
The rate of a chemical reaction is defined as the change in the concentration of reactants or products per unit of time. It measures how quickly a reaction proceeds.
Mathematical Definition
The rate of reaction can be expressed mathematically as:
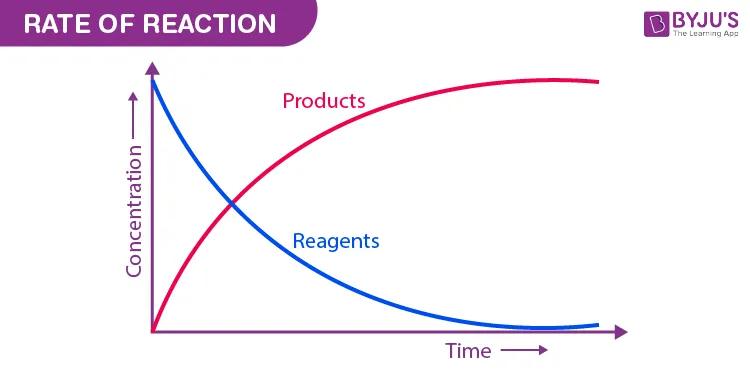
- The concentration of reactants decreases over time.
- The concentration of products increases over time.
Therefore, the rate of a reaction can be defined in two ways:
- The decrease in the concentration of reactants per unit time.
- The increase in the concentration of products per unit time.
The standard unit for reaction rate is mole per cubic decimeter per second ().
Graphical Representation
For a general reaction where a reactant A is converted into a product B:
The change in concentration of reactants and products over time can be visualized graphically.
Instantaneous Rate of Reaction
For very small changes in concentration () over a very small time interval (), the instantaneous rate of reaction is expressed as:
Considering the reaction :
- The rate can be expressed as the rate of disappearance of reactant A.
- The rate can also be expressed as the rate of appearance of product B.
- Where and are the changes in the concentration of A and B.
- The negative sign indicates a decrease in the concentration of the reactant A.
- The positive sign indicates an increase in the concentration of the product B.
This rate expression is fundamental to the study of chemical kinetics and the determination of reaction orders.
Worked Example 7.1
Problem: Determine the average rate of the following reaction, , using the experimental data provided.
The concentration of product C was measured at different time intervals as follows:
| Sr. No. | Time (s) | Concentration of C () |
|---|---|---|
| 1 | 0.0 | 0.0 |
| 2 | 10 | 0.20 |
| 3 | 20 | 0.38 |
| 4 | 30 | 0.45 |
| 5 | 40 | 0.60 |
Solution: Calculate the rate of reaction between the time intervals of 0.0 seconds and 20 seconds.
-
Calculate the change in time ():
-
Calculate the change in concentration of C ():
-
Apply the rate formula:
Note: This calculation gives the average rate of reaction over the specified time interval.
Summary
- Rate of Reaction: The speed at which reactants are converted into products, measured as the change in concentration per unit time.
- Mathematical Expression:
- Units: The standard unit for the rate of reaction is .
- Graphical Interpretation: The slope of a concentration-time graph represents the reaction rate.
- Reactant curve has a negative slope.
- Product curve has a positive slope.
- Average vs. Instantaneous Rate: The rate calculated over a finite time interval is an average rate, while the rate at a specific moment is the instantaneous rate.