5.2 Physical Properties of Liquids
This section explores the key physical properties of liquids, including evaporation, vapour pressure, boiling point, viscosity, and surface tension, with a special focus on how hydrogen bonding influences the unique properties of water.
5.2.1 Evaporation
Evaporation is the spontaneous change of a liquid into its vapour phase at the surface. This process can occur at any temperature.
- Mechanism: Molecules within a liquid have a range of kinetic energies. Those with sufficiently high kinetic energy can overcome the intermolecular attractive forces at the surface and escape into the gas phase.
- Cooling Effect: When the highest-energy molecules escape, the average kinetic energy of the remaining liquid molecules decreases. This results in a drop in the liquid's temperature. To regain thermal equilibrium, the liquid absorbs heat from its surroundings, causing the surroundings to cool down.
Factors Affecting Evaporation:
- Surface Area: A larger surface area allows more molecules to be at the interface, increasing the rate of evaporation.
- Temperature: Higher temperatures increase the average kinetic energy of molecules, meaning a larger fraction of them have enough energy to escape, thus increasing the evaporation rate.
- Intermolecular Forces: Liquids with weaker intermolecular forces (e.g., gasoline, ethyl ether) evaporate faster than those with stronger forces (e.g., water, which has strong hydrogen bonds).
Real-World Application: Why Does High Air Humidity Make it Feel Hotter? Our bodies cool down through the evaporation of sweat. When the air is humid (high concentration of water vapour), the rate of evaporation from our skin slows down significantly. This reduces the body's ability to cool itself, making us feel hotter than the actual air temperature might suggest.
5.2.2 Vapour Pressure
When a liquid is in a closed container, molecules evaporate from the surface and enter the gas phase. Some of these gas-phase molecules (vapours) will collide with the liquid surface and be recaptured in a process called condensation.
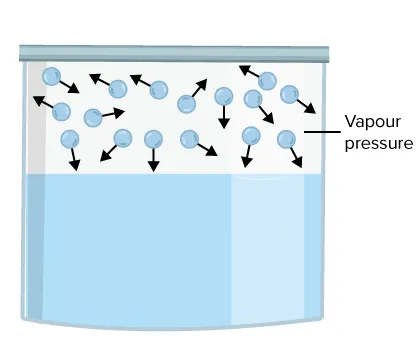
- Dynamic Equilibrium: Initially, the rate of evaporation is greater than the rate of condensation. As more vapour accumulates, the rate of condensation increases until it becomes equal to the rate of evaporation. This state, where the two opposing processes occur at the same rate, is called dynamic equilibrium.
- Definition: Vapour pressure is the pressure exerted by the vapour of a liquid when it is in dynamic equilibrium with the liquid at a given temperature.
Factors Affecting Vapour Pressure:
- Nature of the Liquid (Intermolecular Forces):
- Stronger intermolecular forces hold molecules in the liquid phase more tightly, making it harder for them to evaporate. This results in a lower vapour pressure.
- Weaker intermolecular forces allow molecules to escape more easily, resulting in a higher vapour pressure.
- Temperature:
- Increasing the temperature increases the kinetic energy of the molecules, leading to a higher rate of evaporation and thus a higher vapour pressure. For example, the vapour pressure of water increases from at to at .
Note: Vapour pressure is independent of the amount of liquid, the volume of the container, or the surface area of the liquid.
Table 5.1: Vapour pressure of some liquids at .
| Name of Compound | Vapour Pressure at (torr) |
|---|---|
| Glycerol | 0.00016 |
| Mercury | 0.012 |
| Water | 43.9 |
| Carbon Tetrachloride | 87 |
| Chloroform | 170 |
| Ethyl ether | 442.2 |
| Isopentane | 580 |
5.2.3 Boiling Point
The boiling point is the temperature at which the vapour pressure of a liquid becomes equal to the external (atmospheric) pressure. At this temperature, bubbles of vapour form throughout the bulk of the liquid, and it rapidly converts into a gas.
- Molar Heat of Vaporization (): The amount of heat energy required to convert one mole of a liquid into its vapour at its boiling point. For water, . This energy is used to overcome intermolecular forces, not to increase the temperature, which remains constant during boiling.
Boiling Point and External Pressure: The boiling point of a liquid is directly dependent on the external pressure.
- High External Pressure: More energy is required for the vapour pressure to match the external pressure, so the liquid boils at a higher temperature.
- Example: A pressure cooker increases the pressure inside, raising the boiling point of water to around (at 1489 torr), which cooks food faster.
- Low External Pressure: Less energy is required, so the liquid boils at a lower temperature.
- Example: Atop Mount Everest, where the atmospheric pressure is about 323 torr, water boils at only . In Murree (700 torr), water boils at .
- Vacuum Distillation: By reducing the pressure above a liquid, its boiling point is lowered. This technique is used to distil heat-sensitive substances (e.g., certain organic compounds or pharmaceuticals) that would decompose if heated to their normal boiling point.
5.2.4 Viscosity
Viscosity is the resistance of a liquid to flow. It is a measure of the internal friction between layers of a liquid as they move past one another.
- Cause: Viscosity arises from intermolecular attractive forces. Molecules in adjacent layers of a flowing liquid attract each other, resisting relative motion.
- Effect of Intermolecular Forces: Liquids with stronger intermolecular forces have higher viscosity (e.g., glycerol, honey, motor oil). Liquids with weaker forces have lower viscosity (e.g., water, ethanol).
- Effect of Temperature: Viscosity decreases with increasing temperature because higher kinetic energy allows molecules to overcome intermolecular attractions more easily, enabling layers to flow past each other more freely.
- Effect of Molecular Size and Shape: Larger molecules and those with complex shapes (e.g., long-chain polymers) tend to have higher viscosity due to greater entanglement and surface area for intermolecular interactions.
Water and Viscosity: Water has a relatively high viscosity for a small molecule due to its extensive hydrogen bonding network. However, it is much less viscous than glycerol, which has three groups capable of forming multiple hydrogen bonds per molecule.
5.2.5 Surface Tension
Surface tension is the energy required to increase the surface area of a liquid by a unit amount, or equivalently, the force per unit length acting along the surface. It arises because molecules at the surface experience a net inward force (cohesive force), unlike interior molecules which are attracted equally in all directions.
- Cause: Interior molecules are surrounded by neighbours on all sides and experience balanced intermolecular attractions. Surface molecules have neighbours only below and to the sides, resulting in a net inward pull. This causes the surface to contract to the minimum possible area, behaving like a stretched elastic membrane.
- Effect of Intermolecular Forces: Stronger intermolecular forces → higher surface tension. Water has one of the highest surface tensions of any common liquid due to its strong hydrogen bonding.
Consequences of Surface Tension:
- Small insects (e.g., water striders) can walk on water.
- Water forms spherical droplets (minimum surface area for a given volume).
- A steel needle can float on water if placed carefully.
Effect of Temperature: Surface tension decreases with increasing temperature because higher kinetic energy weakens the cohesive forces at the surface.
5.2.6 Unique Properties of Water Due to Hydrogen Bonding
Water () exhibits several anomalous physical properties that are directly explained by its strong and extensive hydrogen bonding network (each water molecule can form up to 4 hydrogen bonds).
| Property | Value / Observation | Explanation via Hydrogen Bonding |
|---|---|---|
| High boiling point | (unusually high for ) | Strong H-bonds require large energy input to break, raising and boiling point |
| High surface tension | at (highest of common liquids) | Strong cohesive H-bonds create large net inward force at surface |
| High specific heat capacity | Energy absorbed breaks H-bonds rather than raising temperature | |
| High | Extensive H-bond network must be disrupted during vaporization | |
| Low vapour pressure | at | Strong H-bonds reduce tendency of molecules to escape into vapour phase |
| Density anomaly of ice | Ice is less dense than liquid water | In ice, H-bonds form a rigid open hexagonal lattice; upon melting, some H-bonds break and molecules pack more closely |
Biological Significance: Water's high specific heat capacity and high make it an ideal temperature buffer for living organisms and climate regulation. Its high surface tension enables capillary action in plants and supports aquatic surface ecosystems.