3.3 Polar and Nonpolar Covalent Bonds
This section explores the distinction between polar and nonpolar covalent bonds and how the overall polarity of a molecule is determined by both bond polarity and molecular geometry.
Polar Covalent Bonds
A polar covalent bond is formed when there is a significant difference in electronegativity between the two atoms sharing electrons. The more electronegative atom attracts the shared electron pair more strongly, acquiring a partial negative charge (). The less electronegative atom consequently acquires a partial positive charge ().
This separation of charge creates a dipole, which is a pair of equal and opposite electric charges separated by a distance. Molecules with a net dipole are called polar molecules.
Nonpolar Covalent Molecules
A molecule can be nonpolar for two main reasons:
- It consists of only nonpolar bonds (e.g., , ).
- It has polar bonds, but the molecular geometry is symmetrical, causing the bond dipoles to cancel each other out.
Symmetrical Geometries:
Symmetrical polyatomic molecules are often nonpolar. Key symmetrical shapes include:
- Linear
- Trigonal Planar
- Tetrahedral
In these arrangements, the individual bond dipoles exert equal and opposite effects, resulting in a net dipole moment of zero. Therefore, the molecule has no overall charge and is considered nonpolar.
Example: Carbon Dioxide ()
The carbon dioxide molecule provides a classic example of a nonpolar molecule with polar bonds.
- Bond Polarity: There is a significant electronegativity difference between Carbon () and Oxygen (). This makes each Carbon-Oxygen double bond (C=O) polar.
- Molecular Geometry: is a linear molecule (). The two C=O bond dipoles are equal in magnitude but point in opposite directions.
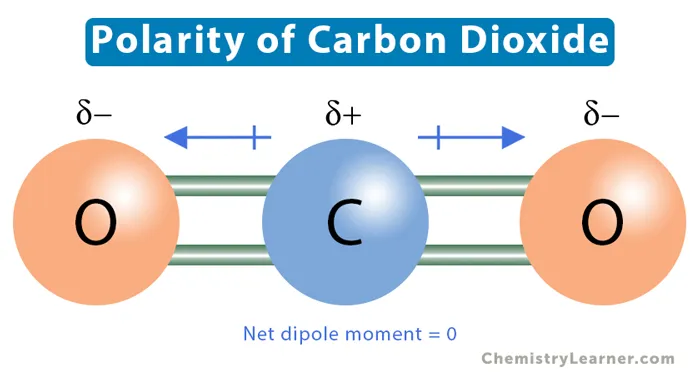
As shown in the diagram, the dipoles cancel each other out, leading to a zero net dipole moment. Thus, is a nonpolar molecule.
Concept Assessment Exercise 3.1
Determine if the following molecules are polar or nonpolar, providing a reason for each.
Given Electronegativity Values: , , , , ,
a. Chlorine,
- Analysis: The bond is between two chlorine atoms, which have the same electronegativity.
- Electronegativity Difference ():
- Conclusion: The Cl-Cl bond is a pure (nonpolar) covalent bond. The molecule is nonpolar.
b. Hydrogen fluoride,
- Analysis: The bond is between hydrogen and fluorine.
- Electronegativity Difference ():
- Conclusion: The H-F bond is highly polar. Since it is a diatomic molecule, there is a net dipole moment. The molecule is polar.
c. Sulfur dichloride,
- Analysis: The molecule has two S-Cl bonds.
- Electronegativity Difference ():
- Conclusion: The S-Cl bonds are polar. The molecule has a V-shaped (bent) geometry, which is asymmetrical. The bond dipoles do not cancel each other out, resulting in a net dipole moment. The molecule is polar.
d. Chloromethane,
- Analysis: The molecule has three C-H bonds and one C-Cl bond arranged in a tetrahedral geometry around the central carbon atom.
- Electronegativity Difference ():
- C-Cl: (Polar)
- C-H: (Slightly polar)
- Conclusion: The molecule is asymmetrical because the atoms bonded to the central carbon are not identical. The strong C-Cl bond dipole is not canceled by the C-H bond dipoles. The molecule is polar.
e. Tetrabromomethane,
- Analysis: The molecule has four C-Br bonds arranged in a tetrahedral geometry.
- Electronegativity Difference ():
- Conclusion: The C-Br bonds are slightly polar. However, the molecule has a symmetrical tetrahedral shape. The four C-Br bond dipoles are arranged symmetrically and cancel each other out. The molecule is nonpolar.