3.1 Electronegativity
Electronegativity is the measure of the ability of a covalently bonded atom to attract the shared pair of electrons towards itself within a molecule.
It is a dimensionless property and has no units because it is a relative tendency, not a form of energy. The most commonly used scale for measuring electronegativity was developed by the American chemist Linus Pauling.
3.1.1 Factors Affecting Electronegativity
Electronegativity is influenced by several key atomic properties:
- Size of the Atom: Smaller atoms have a shorter distance between the nucleus and the valence electron shell. This results in a stronger attraction for the shared electrons, leading to higher electronegativity.
- Nuclear Charge: A higher number of protons in the nucleus results in a greater positive charge. This increased nuclear charge attracts the bonding electrons more strongly, increasing electronegativity.
- Screening by Inner Electrons (Shielding Effect): Inner-shell electrons repel the valence electrons, "shielding" them from the full attractive force of the nucleus. An increase in the number of inner shells leads to a greater shielding effect, which weakens the nucleus's pull on the shared electrons and decreases electronegativity.
In summary:
- As nuclear charge increases and atomic size decreases, the attraction for bonding electrons increases, causing electronegativity to increase.
- As the shielding effect increases, the attraction for bonding electrons decreases, causing electronegativity to decrease.
Table 3.1: Electronegativity of elements in the periodic table
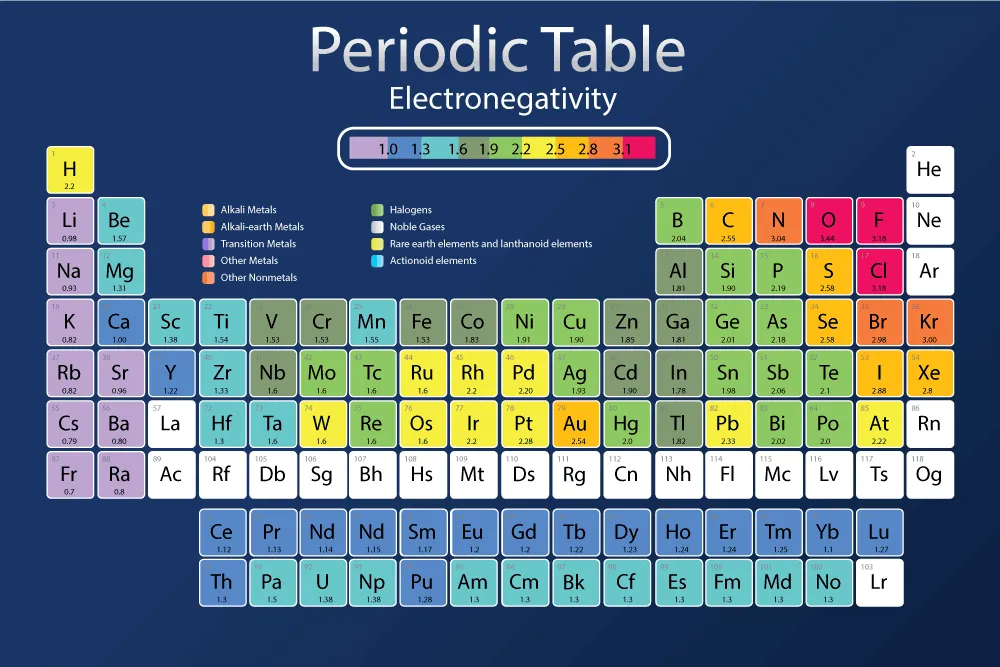
| Li: 0.98 | B: 2.04 | C: 2.55 | N: 3.04 | O: 3.44 | F: 3.98 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Na: 0.93 | Mg: 1.31 | Al: 1.61 | Si: 1.90 | P: 2.19 | S: 2.58 | Cl: 3.16 | |||||||||
| K: 0.82 | Ca: 1.00 | Sc: 1.36 | Ti: 1.54 | V: 1.63 | Cr: 1.66 | Mn: 1.55 | Fe: 1.83 | Co: 1.88 | Ni: 1.91 | Cu: 1.90 | Zn: 1.65 | Ga: 1.81 | Ge: 2.01 | As: 2.18 | Se: 2.55 |
| Rb: 0.82 | Sr: 0.95 | Y: 1.22 | Zr: 1.33 | Nb: 1.60 | Mo: 2.16 | Tc: 1.90 | Ru: 2.20 | Rh: 2.28 | Pd: 2.20 | Ag: 1.93 | Cd: 1.69 | In: 1.78 | Sn: 1.96 | Sb: 2.05 | Te: 2.10 |
| Cs: 0.79 | Ba: 0.89 | La: 1.10 | Hf: 1.30 | Ta: 1.50 | W: 2.36 | Re: 1.90 | Os: 2.20 | Ir: 2.20 | Pt: 2.28 | Au: 2.54 | Hg: 2.00 | Tl: 1.62 | |||
| Fr: 0.70 | Ra: 0.90 |
3.1.2 Trend of Electronegativity in the Periodic Table
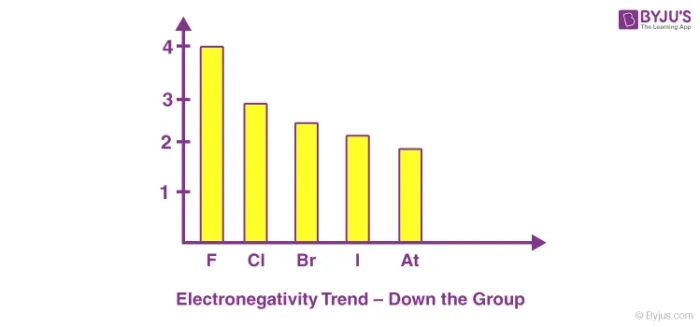
Down a Group
Electronegativity decreases from top to bottom within a group.
- Reason: As you move down a group, a new electron shell is added with each period. This increases the atomic radius and the shielding effect. The increased distance and shielding overcome the effect of the increased nuclear charge, weakening the nucleus's attraction for the shared electrons.
Example: Halogen Group (Group 17)
The electronegativity decreases from Fluorine (F) to Astatine (At). Halogens→
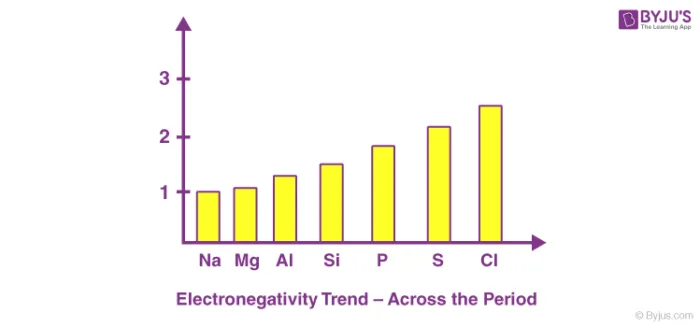
Across a Period
Electronegativity increases from left to right across a period.
- Reason: As you move across a period, protons are added to the nucleus, increasing the effective nuclear charge. The electrons are added to the same principal energy level, so the shielding effect remains relatively constant. The stronger nuclear pull on the electrons decreases the atomic size and increases the atom's ability to attract a shared pair of electrons.
Example: Third Period
The electronegativity increases from Sodium (Na) to Chlorine (Cl).
3.1.3 Nature of Bond in Terms of Pauling Electronegativity Values
The difference in electronegativity () between two bonded atoms determines the nature of the chemical bond.
| Range | Bond Type | Description |
|---|---|---|
| Ionic Bond | One atom effectively donates an electron to the other, forming ions. The electron pair is so unequally shared that the bond is considered ionic. | |
| Polar Covalent Bond | The electron pair is shared unequally. The more electronegative atom acquires a partial negative charge () and the less electronegative atom acquires a partial positive charge (). | |
| Nonpolar Covalent Bond | The electron pair is shared approximately equally. When (identical atoms), the bond is purely covalent. |
Examples:
- : → Ionic bond
- : , between 0.4 and 1.8 → Polar covalent bond
- : → Nonpolar covalent bond