12.3 X-Ray Crystallography
X-ray crystallography is a powerful analytical technique used to determine the three-dimensional (3D) atomic structure of biomolecules such as proteins, DNA, and RNA. It has been fundamental to breakthroughs in medicine, biotechnology, and the understanding of molecular function.
Principle
X-ray crystallography works by scattering a beam of X-rays off the electrons of atoms within a crystallized molecule. The resulting diffraction pattern is analyzed to reconstruct a 3D model of the molecule's electron density, from which an atomic model is built.
The Process of X-ray Crystallography
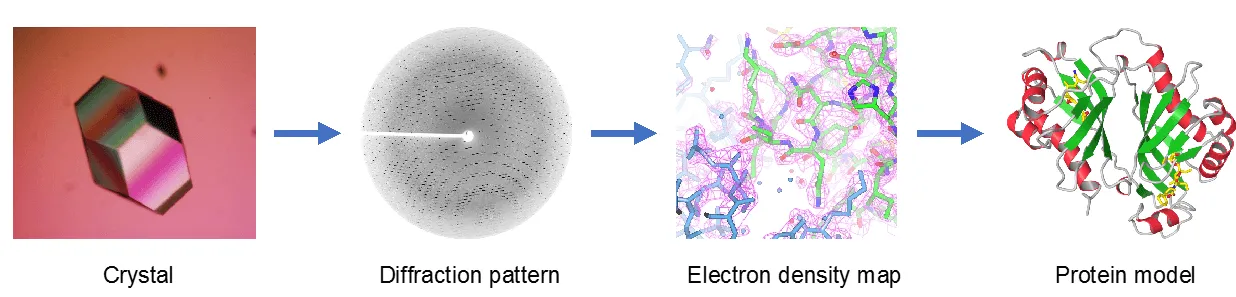
The technique involves a sequential, four-step process to get from a purified biomolecule to a final 3D structure.
Step 1: Crystallization
The target biomolecule must first be purified and induced to form a crystal. This is achieved by dissolving the molecule in a solution and then slowly changing the conditions, such as evaporating the solvent or cooling, to allow the molecules to arrange themselves into a highly ordered, repeating 3D lattice. The quality of the crystal is critical; it must be uniform and have minimal defects for a clear diffraction pattern.
Step 2: X-Ray Diffraction
The crystal is mounted and exposed to a focused beam of X-rays. As the X-rays pass through the crystal, they are scattered or diffracted by the electrons of the atoms in the molecule. The ordered arrangement of molecules in the crystal causes the scattered waves to interfere constructively in specific directions, producing a diffraction pattern of intense spots or reflections. This pattern is recorded by a detector, such as a CCD camera.
Step 3: Data Collection and Processing
The collected diffraction data, consisting of spots and their intensities, are processed using specialized software. Using mathematical techniques, specifically a Fourier transform, the reflection intensities are used to calculate and reconstruct a 3D electron density map of the biomolecule. This map shows the distribution of electrons throughout the crystal's repeating unit, essentially outlining the shape of the molecule.
Step 4: Model Building and Refinement
A molecular model representing the atoms and bonds is manually and computationally fitted into the calculated electron density map. This model is then refined to best match the experimental data. The final result is a precise 3D coordinate file that describes the position of every atom in the molecule, revealing its structure, bond lengths, and angles.
Diagram of the Process
Possible Questions and Answers
Q: Why is crystallization necessary for this technique?
A: A crystal contains a vast number of molecules arranged in an identical, ordered orientation. This regular lattice amplifies the weak scattering signal from a single molecule, producing a sharp, measurable diffraction pattern. Without a crystal, the scattered X-rays would be random and produce no usable pattern.
Q: What information does the diffraction pattern provide?
A: The positions and intensities of the diffraction spots contain the information needed to determine the size of the repeating unit in the crystal and the arrangement of atoms within it. This data is mathematically processed to generate an electron density map.
Q: What is an electron density map?
A: It is a 3D map that shows regions of high and low electron density. Areas of high density correspond to the locations of atoms, since electrons orbit atomic nuclei, allowing scientists to build an atomic model of the molecule.